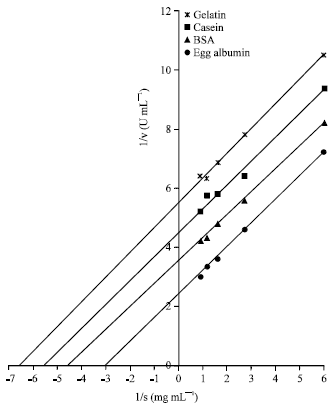
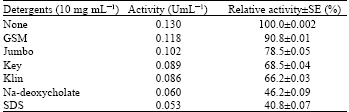
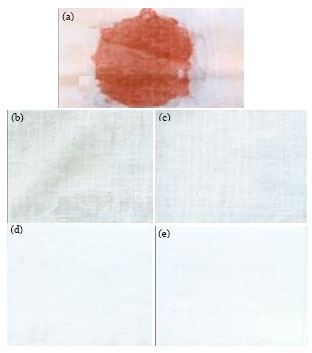
Research Article
Purification and Some Properties of Thermo-stable Alkaline Serine Protease from Thermophilic Bacillus sp. Gs-3
Department of Applied Microbiology and Brewing, Enugu State, University of Science and Technology, Agbani, Nigeria
F. J.C. Odibo
Department of Applied Microbiology and Brewing, Nnamdi Azikiwe University, Awka, Nigeria