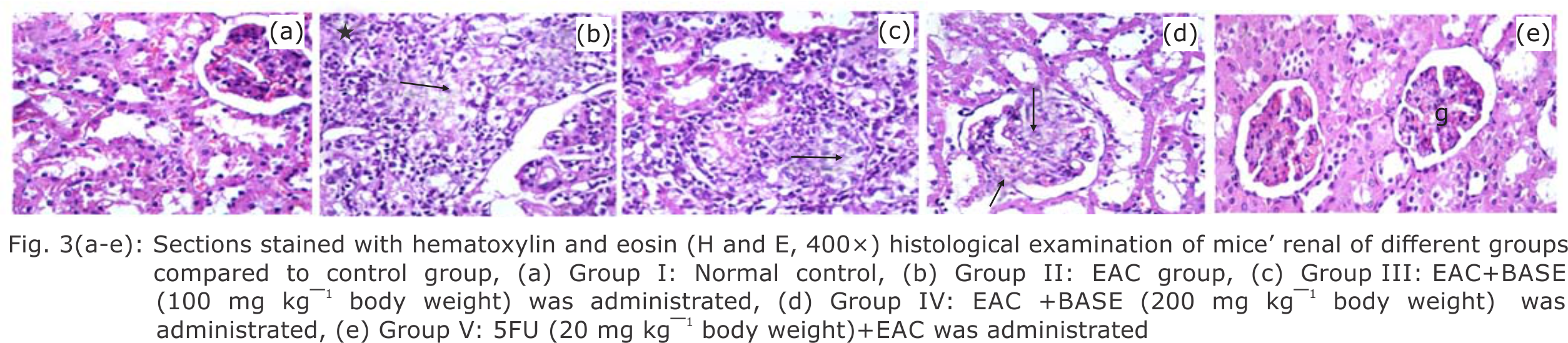
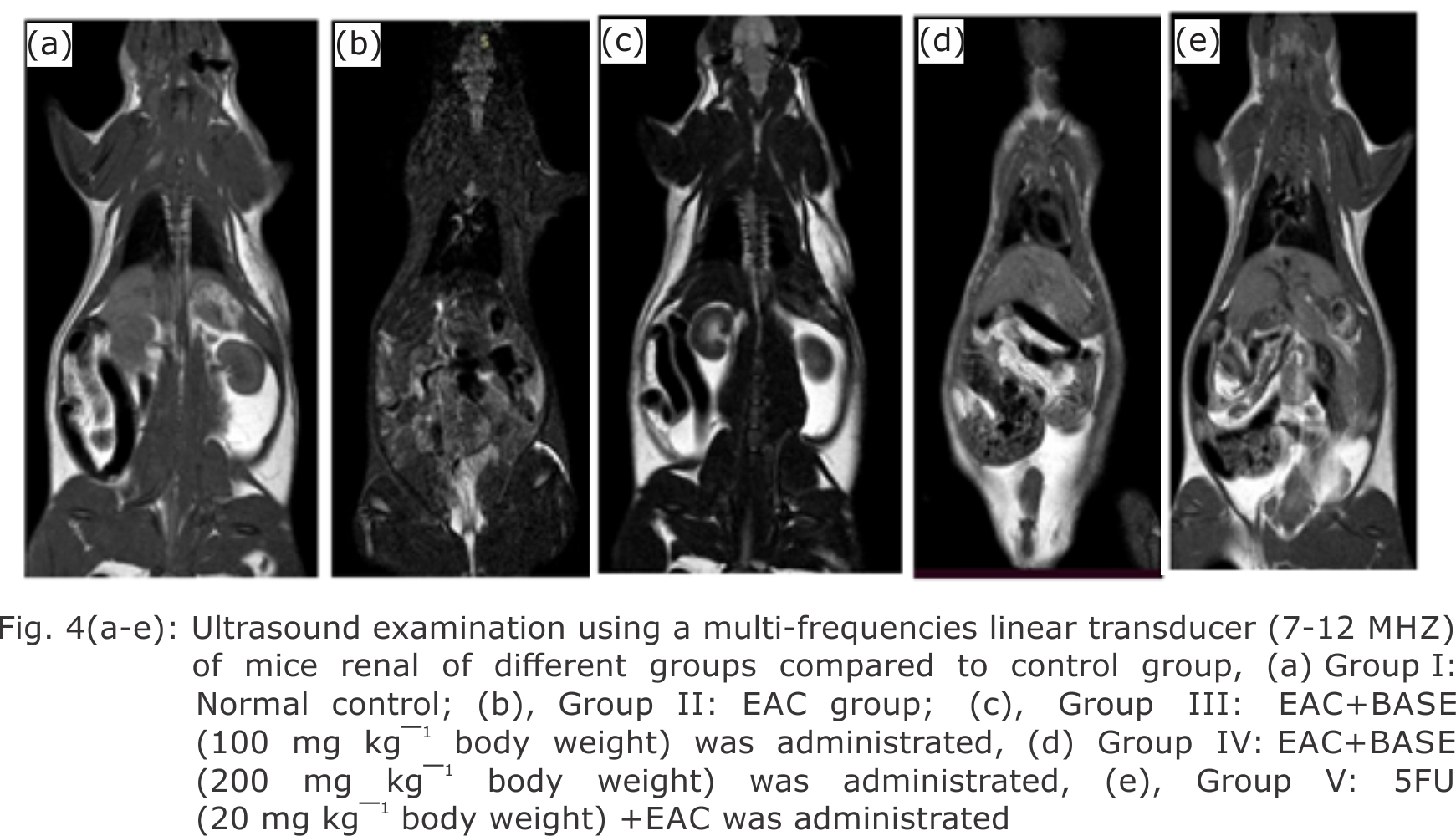
Research Article
Mechanism of Anti-Angiogenic and Renal Protective Activity of Balanites aegyptiaca Seeds Extract in Ehrlich Ascites Carcinoma-Bearing Mice
Department of Radiology and Medical Imaging, Faculty of Applied Medical Science, October 6th University, October 6th City, Egypt
Maraym F. Dawood
Department of Radiology and Medical Imaging, Faculty of Applied Medical Science, October 6th University, October 6th City, Egypt
Wafaa A. Abdallah
Department of Radiology and Medical Imaging, Faculty of Applied Medical Science, October 6th University, October 6th City, Egypt
Aya Z. Hazaa
Department of Radiology and Medical Imaging, Faculty of Applied Medical Science, October 6th University, October 6th City, Egypt
Alyaa A. Ahmed
Department of Radiology and Medical Imaging, Faculty of Applied Medical Science, October 6th University, October 6th City, Egypt
Aya S. Elbayoumy
Department of Radiology and Medical Imaging, Faculty of Applied Medical Science, October 6th University, October 6th City, Egypt
Asmaa M. El-nossairy
Department of Radiology and Medical Imaging, Faculty of Applied Medical Science, October 6th University, October 6th City, Egypt
Nuran S. Yaqoub
Department of Radiology and Medical Imaging, Faculty of Applied Medical Science, October 6th University, October 6th City, Egypt
Shymaa H. Khalifa
Department of Radiology and Medical Imaging, Faculty of Applied Medical Science, October 6th University, October 6th City, Egypt
Elshimaa O. Abdelaziz
Department of Radiology and Medical Imaging, Faculty of Applied Medical Science, October 6th University, October 6th City, Egypt
Abdelrahman B. Khairy
Department of Radiology and Medical Imaging, Faculty of Applied Medical Science, October 6th University, October 6th City, Egypt
Naglaa A. Gobba
Department of Pharmacology, Faculty of Pharmacy, MUST University, October 6th City, Egypt
Esraa A. Abdalla
Department of Biochemistry, Faculty of Applied Medical Science, October 6th University, October 6th City, Egypt
Ismail Hegazy
Department of Biochemistry, Faculty of Applied Medical Science, October 6th University, October 6th City, Egypt
Mohammed A. Hussein
Department of Biochemistry, Faculty of Applied Medical Science, October 6th University, October 6th City, Egypt
LiveDNA: 20.9149