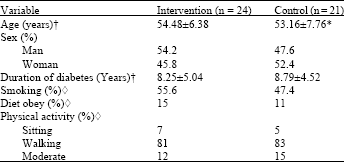
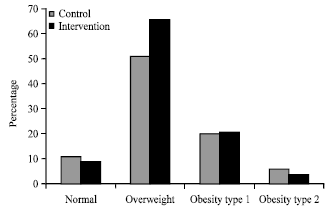
Research Article
The Effect of Hydro Alcoholic Nettle (Urtica dioica) Extract on Oxidative Stress in Patients with Type 2 Diabetes: A Randomized Double-blind Clinical Trial
Nutrition Research Center, Deportment of Nutrition, Faculty of Health and Nutrition, Tabriz University of Medical Science, Iran
A. Tarighat
Nutrition Research Center, Department of Nutrition and Diet Therapy, Faculty of Health and Nutrition, Tabriz University of Medical Science, Iran
A. Bahrami
Endocrinology Unit, Department of Endocrinology, Imam Reza Hospital, Tabriz University of Medical Science, Iran