ABSTRACT
The biological function of colostrums in all lactating species is the growth and health of a newborn offspring. In bovine colostrum, the naturally occurring antibodies (immunoglobulins) provide a marked antimicrobial effects against a wide range of microbes and confer a passive immunity until the calf’s immune system has matured. The concentration of specific antibodies in mammary secretions against a certain pathogens microorganism can be raised in colostrum and milk by immunizing cows with these pathogens or their antigens. Such hyperimmune colostrum and milk or antibodies isolated from it provide an increased specific protection against different enteric diseases in calves. Colostral immunoglobulin preparations designed for farm animals are commercially available in many countries and also some colostrum based products are marketed for humans as dietary supplements. Clinical studies are now in progress in many countries to evaluate the potential of immune milk products as preventative treatment for various hospital infections, especially those caused by antibiotic resistant bacteria and Helicobacter pylori, the causative agent of chronic gastritis. This article reviews the recent progress made in the properties of bovine immunoglobulins, their isolation from colostrum and utilization in the preparation of bovine immune milk for prevention and treatment of microbial infections in humans and animals.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/ijds.2007.183.195
URL: https://scialert.net/abstract/?doi=ijds.2007.183.195
INTRODUCTION
Immunoglobulins (Igs), also called antibodies, are present in the blood and tissue fluids, as well as many secretions, e.g., colostrum and milk of all lactating species. The biological function of milk Igs is to give the offspring an immunological protection against microbial pathogens and toxins and to protect the mammary gland against infections. Igs are divided into classes having different physiochemical structure and biological activities. The major classes in bovine and human milk are IgG, IgM and IgA. The concentration of different Ig classes in colostrum and milk varies considerably according to species, breed, age, stage of lactation and health status and is often different from that in blood. For example, in human colostrum and milk, the IgA class comprises about 90% and in blood 15-20% of total Igs whereas in cow the IgG class is dominant in colostrum, milk and blood (about 80-90, 60-70 and 90% of total Igs, respectively). The transport of Igs from blood serum to milk is a selective process favouring, in most species, homologous IgG. Specific receptors are involved in the process enabling the characteristic concentration of Ig isotypes in colostrum and milk of different species. In many species, the absorption of Igs from intestine is also selective and receptor-mediated. However, in ruminants, e.g., in the cow, the absorption of Igs takes place nonselective during the first 12-36 h after birth of the offspring. Ruminant neonates are born virtually without Igs and the colostral Igs are essential for survival. Thus in ruminants the colostrum contains remarkably higher amounts of Igs than the mature milk. The Ig fractions of milk is used commercially as feed supplements and replaces of colostrum, mainly for neonate calves and pigs in order to prevent gastrointestinal infections.
Increasing interest has recently been focused on the development of colostrum-based Ig products, which contain specific antibodies for the prevention, or treatment of infections in humans (Marnial and Korhonen, 2002a).
Colostral bovine Igs has the potential to be utilized as immunological supplements to infant formula and other hyperimmune foods (Dominguez et al., 1997).
If the animal is immunized by a vaccination protocol against a certain pathogen (or antigenic structure derived from it), the specific antibody titer of colostrum or milk may increase to 100-400 times of that in colostrum or normal milk (Marnial and Korhonen, 2002a).
Immune milk hyper is a generic termed coined by Ehrlich in the 1890’s for the pregnant cows during the dry period that have been immunized by antigens from pathogens, in order to raise specific antibodies in their colostrum and milk (Ehrlich, 1892).
Peterson and Campbell (1955) reintroduced the idea of use immune milk from cows for controlling human disease. Campbell and Peterson (1963), suggest that orally administered bovine colostrum could provide passive immune protection for humans. Since 1980, an increasing number of studies have shown that the immune milk preparation, basically, the approaches to the development of Ig-based preparations are either concentration or isolation of Igs that occur naturally in colostrum, milk and cheese whey (El Loly, 1996; El Loly and Farrag, 2006), or hyper immunization of pregnant cows during the dry period with antigens from pathogens, in order to raise specific antibodies in the colostrum and milk, can be effective in the prevention or treatment of human and animal diseases caused by entropathogeic microbes (Hoerr and Bostwick, 2002).
The development of immune milk products remained for a long time constrained by the lack of appropriate technologies for concentration of Igs from colostrum or milk. Rapid progress made in the membrane separation and chromatographic techniques since the late 1980s has, made it possible to enrich Igs and other individual protein fractions on a large scale from both colostral and cheese whey. This technological progress in parallel with the advent of the health promoting functional foods in the early 1990s has opened up new opportunities for the development of commercial immune milk products. A few products are already on the market and more applications can be expected in the coming years. Biotechnological specialist is committed to become a leading developer and producer of immune milk products for use in the fight against hospital infections, one of health care’s growing problems worldwide. This products offer a novel means for helping patients who cannot, or should not, be treated using current antibiotics (Maubois and Ollivier, 1997; Maubois, 1998; Kelly and McDonagh, 2000).
The relationship between human health and proper nutrition is now generally recognized and publications abound on this subject. Since the late 1970’s much research has demonstrated the crucial role of the nutritional states of hospitalized patients. An intact and well functioning intestinal tract is key to a faster recovery, reduction of complications etc. (Thompson, 1994; Spiller, 1997).
The ecological environment for the luminal microbial flora in the small intestine and colon is of particular importance. A balanced microflora is a major building block for the protective system for humans and animals. Other determination in the protective system against invading pathogenic bodies (microorganisms and endotoxins) is the various components that together from the mucous adjacent to the intestinal epithelial cell-layer. It is a very complex and fragile system, which may become unbalanced or dysfunction by a large number of factors such as: malnutrition, gastrointestinal infections, chemotherapy etc. Such dysfunction may result in one or more of the following clinical symptoms: diarrhea, inflammations, sepsis etc. These may ultimately develop into a life-threatening situation or cause extended illness and/or prolonged hospital stay at substantially increased costs. To these may be added the loss of income and related social costs (Bengmark, 1996, 1999; Wesley, 1990; Campieri and Gionchetti, 2000).
DEFINITION OF IMMUNOGLOBULINS
Immunoglobulins (Igs) are the major components of humoral immune response. As they migrate with the globular proteins in serum electrophoresis, they are named antibodies too. They consist of 20% of total plasma proteins. Antibodies are glycoprotein molecules which are produced by plasma cells in response to an immunogen/antigen. Upon activation, the B cell undergoes clonal selection to produce a clone of effector cells known as plasma cells, which synthesize antibodies specific for the antigenic determinant in question. After binding specifically to the target antigenic determinant, different effector mechanisms are triggered and the antigen is destroyed. Thus the antibody molecules show great heterogeneity (diversity). The antibodies which are derived from more than one clone of B cells are called polyclonal antibodies. In contrast, the antibodies originating from a single B cell clone are homogenous and called monoclonal antibodies. Monoclonal antibodies have been first identified in the patients with multiple myeloma, a plasma cell malignancy. The immunoglobulins are made of 90% of glycoproteins and 10% of carbohydrate, the carbohydrate content ranges from 2-3% for IgG to 12-14% for IgM and IgA (www.path.cam.ac.uk/~mrc7/mikeimages.html).
The nomenclature of the immunoglobulins is based on their immunological cross reaction with reference proteins, preferably of human origin and proposed by the World Health Organisation (Butler, 1971, 1983).
STRUCTURE OF BOVINE IMMUNOGLOBULINS
Igs are present in the colostrum and milk of all lactating species; Igs are divided into classes having different physiochemical structures and biological activities. The major classes in bovine and human lacteal secretions are IgG, IgM and IgA. Igs are made 90% of protein and 10% of carbohydrate. The basic unit (monomeric structure) of all Igs is similar to IgG, which is composed of four polypeptide chains; two identical light chains (23 kDa) and two identical heavy chains (53 kDa) (Fig. 1a). Their two types of light chains (κ and λ) differing in chain structure, the approximate length of light chain is from 211 to 217 amino acids. Each of light chain has a constant region and variable region. The variable region determines the immunological specificity. Light chains are joined to the heavy chains by disulfide bonds and the two heavy chains are held together by disulfide bonds near a hinge region, which it gives the molecule the flexibility needed in antibody-antigen interactions. While heavy chains contain five types (γ, δ, α, μ and ε), they define classes of Igs. Heavy chains α and γ have approximately 450 amino acids, while μ and ε have approximately 550 amino acids. Each heavy chain has a constant region, which is the same by all Igs of the same class and a variable region, which differs between Igs of different B cells, but does the same B cell produce the same for all Igs. Heavy chains γ, α and κδ have the constant region composed of three regions, the constant region of heavy chains μ and ε is composed of four regions. Fc fragment, the stem of the Y is composed of two heavy chains that each contributes two to three constant regions (depending on the class of the antibody). Fc binds to various cell receptor and complement proteins, it mediates different physiological effects of antibodies (opsonization, cell lysis and other processes). The variable region of any heavy chain is composed of one region. These regions are about 110 amino acids long. There are also some amino acids between constant regions. The complete Ig or antibody molecule possesses sterochemically a Y-shaped structure and has a molecular weight of about 160 kDa. Each Ig molecule has two identical antigen-binding sites termed F(ab’)2, which are formed by the N-terminal part of one heavy and one light chain. The C-terminal region of the heavy chains is termed the Fc region. The bovine IgG molecule occurs predominantly, which make up 80-90% of the total Igs in colostrum, in two subclasses: IgG1 and IgG2. The basic structure of monomeric IgM and IgA is similar to that of IgG, except for the addition of a C-terminal octapeptide to the heavy chains. IgA occurs as a monomer or dimmer, the latter comprising two IgA molecules joined together by a J-chin and a secretory component. This complex, called secretoy IgA (SIgA) has a molecular weight of about 380 kDa. Except for ruminant lacteal secretions, IgA is the dominating Igs in all external secretions of the body.
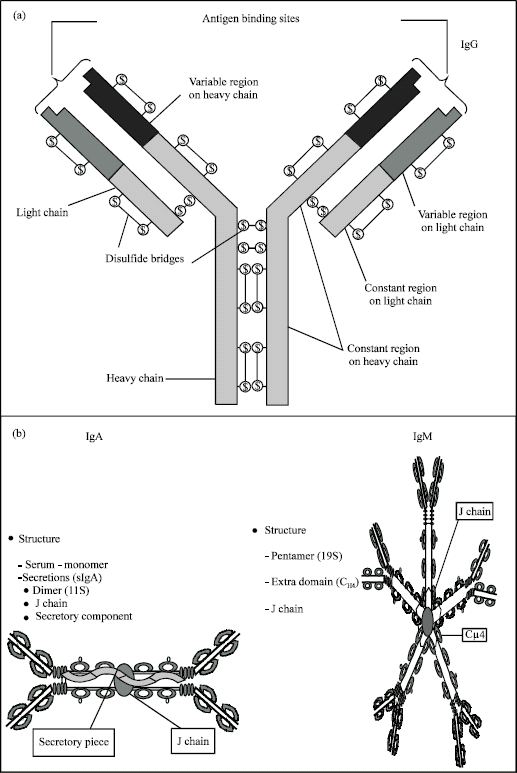 | |
| Fig. 1: | Schematic diagram of structure IgG, IgA and IgM. (www.path.cam.ac.uk/~mrc7/ mikeimages.html) |
IgM consist of five subunits, similar to monomeric IgA, which are linked together in a circular mode by disulfide bonds and a J-chain, the molecule weight of pentameric IgM is approximately 900 kDa (Fig. 1b) (Larson, 1992; Butler, 1974, 1998; Korhonen et al., 2000a; Marnila and Korhonen, 2002a).
| Table 1: | Concentration of immunoglobulins in bovine colostrum and milk* |
 | |
| *Data from references Butler, 1994; Marnial and Korhonen, 2002a | |
Cows secrete colostrum for the first five to seven days after delivery of the calf. The concentration of the various Igs in bovine serum and lacteal secretion varies according to the breed, age, health status and stage of lactation of the animal. Igs make up (70-80%) of the total protein content of colostrum, whereas in mature milk they account for only (1-2%) of total protein (Larson, 1992; Marnial and Korhonen, 2002b).
The concentration of Igs varies considerably (20-200 g L-1) in the first milking after calving being on an average about (60 g L-1). IgG1 is the predominate class comprising over (75%) of the Igs in colostral whey followed by IgM, IgA and IgG2, respectively (Table 1). Total Igs levels in milk decline rapidly following parturition to around (0.7-1.0) g L-1. IgG1, however, remains the predominant subclass in these secretions (Marnial and Korhonen, 2002a).
ISOLATION OF IMMUNOGLOBULINS FROM COLOSTRUM
Traditionally, the globulin fraction was isolated from colostral whey by precipitation with either ammonium sulphate or ethanol. Although these chemical methods yield rather pure Igs fraction they are not appropriate for large-scale isolation of Igs from cow’s colostrum or milk. Since the 1980’s, the rapid development of membrane separation and chromatographic techniques has made it possible to isolate or concentrate Igs from bovine colostrum and milk on an industrial scale. In particular, there has been considerable commercial interest in the isolation of biologically active whey proteins, such as Igs, lactoferrin, lactoperoxidase and glycomacropeptide. A great number of patented pilot-or industrial-scale methods, based on membrane separation and/or chromatographic techniques have been developed over the past 10-15 years (Maubois and Ollivier, 1997; Maubois, 1998; Kelly and McDonagh, 2000).
Among whey proteins, the Igs are molecules with the biggest but highly variable molecular weight. Membrane separation technologies, in particular ultrafiltration (UF), have been employed alone or in combination with ion exchange or gel chromatography for the isolation and purification of Igs from colostral or cheese whey (Abraham, 1988; Kothe et al., 1987; Stott and Lucas, 1989; Syvaoja et al., 1994).
A combination of different membrane technologies has proved to be the most cost-effective approach for the commercial production of crud Igs preparations. Using these methods, the recovery rate of Igs has varied from 40-70% of the concentration present in the starting material. For the improvement of the recovery rate of Igs from whey and to increase the Igs concentration of the final preparation, specific chromatographic techniques need to be applied. For example, IgG was separated successfully from the UF-concentrated whey using immobilized metal chelate chromatography (Al-Mashiki et al., 1988; Fukumoto et al., 1994). For the isolation of bovine IgG subclasses IgG1 and IgG2 (Akita and Li-Chan, 1998) developed an immunoaffinity chromatography process using immobilized egg yolk antibodies.
Korhonen et al. (1998) applied various membrane separation methods (UF, microfiltration and reverse osmosis) and cation exchange resin as a molecular sieve for concentrating Igs from colostral whey. Examples of these processes are outlined in Fig. 2. The Igs level (w/w) of the final freeze-dried concentrates varied from 45 to 75%.
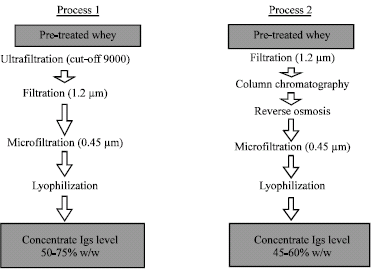 | |
| Fig. 2: | Examples of processes designed for manufacturing of Igs concentrates from bovine colostrums |
Colostrum is the richest source of Igs per volume unite, cheese whey is often considered a more reliable source of these component, since it is available in large volumes despite its relatively low Igs content. On the other hand, colostrum provides physiologically a superior source of specific antibodies when developing immune milk products.
TRANSFER OF IMMUNOGLOBULINS
Immunoglobulins represent the mother’s specific immune response to the antigens encountered in the past. This immunological memory is transported from the mother to the offspring either in utero or via colostrums or milk. In the in utero pathway, Igs pass from the mother’s blood to the fetus across the placenta. In transfer via colostrum or milk the Igs that are derived from blood or are synthesized in the mammary gland are transported through mammary secretory cells into the colostrum or milk. A vast majority of IgG in bovine colostrum and milk is derived from blood plasma. The Igs are produced by B-lymphocytes. Some Igs are carried on the surface of B lymphocytes where they act as receptors and the others are free in body fluids. Contact between B-lymphocytes and antigen is needed to cause the B-lymphocytes to develop into antibody-forming and secreting plasma cells. These cells produce mainly IgA but IgM and IgG are synthesized also to a small extent. The passage of ingested Igs across intestinal cells of the neonate can be specific or nonspecific, depending on the species and it occurs only for a certain period after birth. The gut of the suckling calf, pig or foal is nonselective and open to the transport of a variety of macromolecules for a period of 12-36 h after birth, matching with the time of the highest IgG content of colostrum. The absorption of ingested Igs from the intestine is selective in many species, which transfer the Igs both in utero and via colostrums after birth. Also in the human infant, the colostral Igs are absorbed in small quantities for a short period of 18-24 h after birth (Marnial and Korhonen, 2002a).
DIGESTION OF IMMUNOGLOBULINS
Proteolysis enzymes in the stomach and intestine into small peptides and amino acids, which are subsequently absorbed, normally degrade antibodies ingested by humans. Orally administrated bovine milk Igs are degraded in the human gastrointestinal tract by the intestinal proteases pepsin, trypsin, chmotrypsin, carboxypeptidase and elastase into F(ab’)2, Fab and Fc fragments (Fig. 3). The Y-shaped monomer has two heavy and two light chains. Together this gives six to eight constant regions and four variable regions. Each half of the forked end of the Y is called a Fab fragment. It is composed of one constant and one variable region of each the heavy and the light chain, which together shape the antigen binding site at the amino terminal end of the monomer.
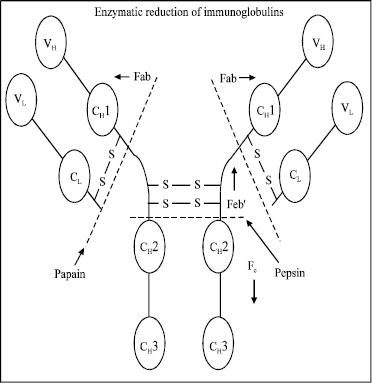 | |
| Fig. 3: | Enzymatic reduction of immunoglobulins (www.path.cam.ac.uk/~mrc7/ mikeimages.html). |
The two variable regions bind their specific antigens (Marnial and Korhonen, 2002a).
The enzyme papain cleaves a monomer into two Fab (fragment antigen binding) fragments and an Fc (fragment crystallizable) fragment. While the enzyme pepsin cleaves below hinge region, so an F(ab’)2 fragment and an Fc fragment is formed. The F(ab’)2 and Fab fragments retain some of the neutralizing and adhesion-inhibiting activities of the intact antibody. The secretory piece component of the IgA molecule makes it more resistant to the activities of proteolytic digestive enzymes than in the case of other Igs classes (Reilly et al., 1997).
In humans, this property is manifested by the passage of a considerable proportion (20-80%) of undigested active IgA from human colostrum through the gut of the neonate baby (Goldman, 1993). In comparison, it has been shown in many in vitro studies that bovine IgG is also relatively resistant to proteolysis by digestive enzymes and is only partially inactivated by gastric acid (Korhonen et al., 2000a). Also, studies with healthy human subjects have shown that a significant amount (about 19%) of the orally administered bovine IgG and IgM can survive the passage through the gastrointestinal tract (Roos et al., 1995). In another human study, some 49% of ingested 2.1 g of bovine colostral Igs was recovered in the ileal fluid of adult volunteers (Warny et al., 1999). In addition, from 10-30% of orally administered bovine Igs can be recovered from the stools of human infants and adults. These recovered Igs are in the form of F(ab’)2, Fab and Fc fragments and retain some immunological activity (Marnial and Korhonen, 2002a). Recovery rates of undigested Igs can be improved considerably by enteric encapsulation of Igs concentrates (Kelly et al., 1997) or fasting before the administration of Igs (Muntjewerf and Korhonen, 2002).
FUNCTIONS OF IMMUNOGLOBULINS
Igs function as flexible adaptors linking together various parts of the cellular and humoral immune systems. While one part F(ab’)2 of an antibody molecule binds to antigen, other parts (mostly the Fc region) interact with other elements. In addition to antigen binding, all Igs exhibit one or more effectors functions, which are capable of preventing the adhesion of microbes to surfaces, inhibiting bacterial metabolism by blocking enzymes, agglutinating bacteria and neutralizing toxins and viruses. Another vital function of Igs is their ability to augment the recognition and phagocytosis of bacteria by leukocytes (opsonization). The immunological function mediated by Igs depends on their class. IgG antibodies, which make up 80-90% of the total protein content in colostrum, provide a multitude of functions, including fixation of complement, opsonization, agglutination of bacteria and neutralizing toxins and viruses. IgM antibodies, although produced in smaller amounts than IgG, are considerably more efficient than IgG with regard to most of the mentioned activities, especially complement-mediated lyses. In contrast, the IgA antibodies, does not fix complement or opsonize bacteria, but agglutinates antigens, neutralizes bacterial toxins and viruses and prevents the adhesion of enteropathogenic bacteria to mucosal epithelial cells (Marnial and Korhonen, 2002a).
Utilization of Immunoglobulins
Technological Properties of Immunoglobulins
Milk Igs may affect many dairy technological processes. Igs are known to inhibit the renneting process in the cheese manufacture if the Igs level of milk is increased, e.g., by adding colostrum or in the case of mastitic milk. The antimicrobial properties of Igs may inhibit fermentation by dairy starters and cause false positive results in antibiotic detection assays based on microbial growth. Furthermore, Igs contribute to cream formation by agglutinating fat globules. This property can be inactivated by pasteurization and homogenization of milk (IDF, 1991).
The effects of processing and storage conditions on the stability of purified Igs or Ig concentrates have been the subjects of many recent studies. During processing, the stability of the Igs activity in colostrum or in milk is influenced by thermal treatments (Lindstrom et al., 1994; Li-Chan et al., 1995; Dominguez et al., 1997). Following high temperature/short time (HTST) pasteurization (72°C/15 sec) or batch pasteurization (63°C/30 min) only 0.5-10% of the Igs activity is lost, whereas ultrahigh temperature (UHT) treatment (138°C/4 sec) destroys the majority of the antibodies activity due to denaturation of Igs molecules, whereas a considerable amount of the antibody activity is retained after spray-drying at low temperatures. The evaporation process appears to fully destroy the specific immune activity of milk (Kummer et al., 1992; Li-Chan et al., 1995). The rapid heat inactivation of IgG starts at temperatures higher than 65°C and at 81°C, as much as 90% of the virus neutralization activity is lost in less than 2 min (Mainer et al., 1999). However, bovine IgG added to UHT milk has been shown to retain its specific immune activity for over five months when stored at different temperatures up to 35°C (Fukumoto et al., 1994; Virtanen et al., 1998). Other technological processes, such as freezing and storage as frozen do not destroy any of the antibody activity. Also, Igs molecules seem to retain their specific activity well in the milk or whey powder, irrespective of the storage temperature. The storage of freez-dried Igs concentrate from colostral whey at 4, 20 and 37°C has been found to have little effect on the immune specificity for up to 12 months of storage (Husu et al., 1993). Moreover, the fermentation of milk by yogurt starter or probiotic bacteria does not reduce significantly the activity of added colostrum-derived antibodies and, the antibodies do not interfere with the fermentation process (Wie et al., 2002).
Applications of Immunoglobulins
Commercial colostral whey-derived Igs preparations have been on market for a long time as feed supplements or substitutes for newborn farm animals (Scammell, 2001). The efficacy of these non-specific Igs products in the prevention and treatment of gastrointestinal infections has proven variable in animal studies (Mee and Mehra, 1995; Garry et al., 1996). On the other hand, Hilpert et al. (1987), describes the immunization of cows with specific antibodies to produce hyper-immune milk, also, documented the collection of milk from cows immunized with the E. coli vaccine to prepare a whey protein concentrate used to protect young animals from disease.
| Table 2: | Efficacy of bovine immune colostrum or milk against microbial infections in humans |
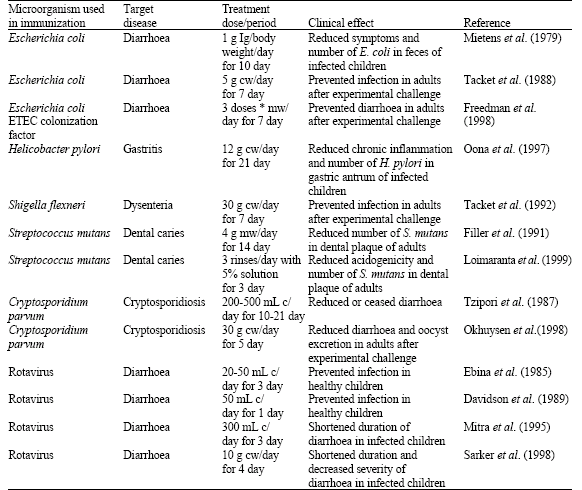 | |
| Ig: Immunoglobulin, cw: Colostral whey concentrate, mw: Milk whey concentrate, c: Colostrum,*: Size not indicated | |
Korhonen et al. (2000a), found that the ample evidence that Ig concentrates or purified Igs isolated from colosrum or milk of hyperimmunized cows provides protection against diarrhea in calves. Li-Chan et al. (1994), found increased ELISA activity for only one of five bacterial antigen in immunized cows in comparison to the levels of activity in milk from commercial dairies.
Over the last 15 years, an increasing number of controlled clinical studies have shown that the oral administration of immune milk preparations containing high titers of specific antibodies can provide effective protection and to some extent may also be therapeutic value against gastrointestinal infections in humans (Korhonen et al., 2000a; Weiner et al., 1999; Lilius and Marnila, 2001).
Several commercial immune-milk products with specific antibodies are available in the market (Pakkanen and Aalto, 1997). They include Gastrogard (Northfield Laboratories, Oakden, Australia), a product used to prevent diarrhea caused by rotavirus in young children and PRO-IMMUNE 99 (GalaGen Inc., Minnesota, USA), a product used on young calves to prevent scours caused by E. coli. Furthermore, Biotest Pharm GmbH (Frankfurt, Germany) produces Lactimmunoglobulin Biotest, a product for human subjects, which contains immunoglobulins from colostrum of non-immunized cows. It has been tested in the treatment of severe diarrhea in AIDS patients (Stephan et al., 1990).
Viable byproducts Ltd. (Turku, Finland) produces Bioenervi, a sterile-filtered colostrum-based product, which is designed to provide growth and antimicrobial factors during strenuous physical activity, e.g., training of athletes (Mero, 1995).
Table 2 presents examples of recent clinical human studies in which specific immune milk preparations have been employed. Many immune milk preparations are being tested in clinical studies for their efficacy, as reviewed by Muntjewerf and Korhonen (2002).
CONCLUSIONS
Isolation of immunoglobulins from bovine colostrum and milk has enabled the development of commercial immunoglobulin preparations targeted at health promotion in farm animals and humans. In order to improve the efficacy of these preparations customized immunization schemes of cows has been designed with the purpose of raising specific antibodies. The enhancement of specific antibody activities of milk immunoglobulins through targeted immunization and isolation/concentration technologies may in future have remarkable potential in producing immunoglobulin products for human health care, not only as part of a health-promoting diet, but also as an alternative or supplement to the medical cure of specified human diseases.
Colostral or milk immunoglobulins can prevent the attachment of pathogen to the epithelial lining that is a critical step in the establishment of infection. Thus, orally administered bovine colostral or milk immunoglobulins have proven effective in the prevention of orally mediated infections.
FUTURE PROSPECTS
According to Korhonen (1998), immune milk might be targeted to specific consumer groups in the future.
More applications can be expected in the coming years, as immune milk products are considered as promising examples of health-promoting functional foods or nutraceuticals.
REFERENCES
- Akita, E.M. and E.C. Li-Chan, 1998. Isolation of bovine immunoglobulin G subclasses from milk, colostrum and whey using immobilized egg yolk antibodies. J. Dairy Sci., 81: 54-63.
Direct Link - Al-Mashikhi, S.A., E. Li-Chan and S. Nakai, 1988. Separation of immunoglobulins and lactoferrin from cheese whey by chelating chromatography. J. Dairy Sci., 71: 1747-1755.
CrossRefDirect Link - Bengmark, S., 1996. Econutrition and health maintenance: A new concept to prevent GI inflammation, ulceration and sepsis. Clin. Nutr., 15: 1-10.
Direct Link - Bengmark, S., 1999. Gut microenvironment and immune function. Curr. Opin. Cin. Nutr. Metab. Care, 2: 83-85.
PubMedDirect Link - Butler, J.E., 1983. Bovine immunoglobulins: An augmented review. Vet. Immunol. Immunopathol., 4: 43-152.
CrossRefPubMedDirect Link - Butler, J.E., 1998. Immunoglobulin diversity, B-cell and antibody repertoire development in large animals. Rev. Sci. Tech. Off. Int. Epi., 17: 43-70.
PubMedDirect Link - Dominguez, E., M.D. Perez and M. Calvo, 1997. Effect of heat treatment on the antigen-binding activity of antiperoxidase immunoglobulins in bovine colostrum. J. Dairy Sci., 80: 3182-3187.
Direct Link - Ebina, T., A. Sato, K. Umezu, N. Ishida and S. Ohyama et al., 1985. Prevention of rotavirus infection by oral administration of cow colostrum containing anti-human rotavirus antibody. Med. Microbiol. Immunol., 174: 177-185.
CrossRefPubMedDirect Link - Freedman, D.J., C.O. Tacket, A. Delehanty, D.R. Maneval, J. Nataro and J.H. Crabb, 1998. Milk immunoglobulin with specific activity against purified colonization factor antigens can protect against oral challenge with enterotoxigenic Escherichia coli. J. Infect. Dis., 177: 662-667.
PubMedDirect Link - Garry, F.B., R. Adams, M.B. Cattell and R.P. Dinsmore, 1996. Comparison of passive immunoglobulin transfer to dairy calves fed colostrum or commercially available colostral supplement products. JAVMA, 208: 107-110.
PubMedDirect Link - Hoerr, R.A. and E.F. Bostwick, 2002. Commercializing colostrum-based products: A case study of GalaGen. Inc., Bull. Int. Dairy Fed., 375: 33-46.
Direct Link - Kelly, C.P., S. Chetman, S. Keates, E. Bostwick and A.M. Roush et al., 1997. Survival of anti-Clostridium difficile bovine immunoglobulin concentrate in the human gastrointestinal tract. Antimicrobial Agents Chemother., 41: 236-241.
Direct Link - Korhonen, H., 1998. Colostrum immunoglobulins and the complement system-potential ingredients of functional foods. IDF Bull., 336: 36-40.
Direct Link - Korhonen, H., P. Marnila and H.S. Gill, 2000. Milk immunoglobulins and complement factors. Br. J. Nutr., 84: S75-S80.
CrossRefPubMedDirect Link - Korhonen, H., P. Marnila and H.S. Gill, 2000. Bovine milk antibodies for health. Br. J. Nutr., 84: S135-S146.
PubMedDirect Link - Li-Chan, E., A. Kummer, J.N. Losso, D.D. Kitts and S. Nakai, 1995. Stability of bovine immunoglobulins to thermal treatment and processing. Food Res. Int., 28: 9-16.
CrossRefDirect Link - Lilius, E.M. and P. Marnila, 2001. The role of colostral antibodies in prevention of microbial infections. Curr. Opin. Infect. Dis., 14: 295-300.
PubMedDirect Link - Loimaranta, V., M. Laine, E. Sderling, E. Vasara and S. Rokka et al., 1999. Effects of bovine immune and non-immune whey preparations on the composition and pH response of human dental plaque. Eur. J. Oral Sci., 107: 244-250.
CrossRefDirect Link - Mainer, G., E. Dominguez, M. Randrup, L. Sanchez and M. Calvo, 1999. Effect of heat treatment on anti-rotavirus activity of bovine immune milk. J. Dairy Res., 66: 131-137.
CrossRefDirect Link - Mitra, A.K., D. Mahalanibus, H. Ashrof, L. Unicomb, R. Eeckels and S. Tzipori, 1995. Hyperimmune cow colostrum reduces diarrhoea due to rotavirus: A double-blind, controlled clinical trial. Acta Paediatr., 84: 996-1001.
PubMedDirect Link - Muntjewerf, A.K. and H. Korhonen, 2002. Bovine immunoglobulins: Technological and applications. Bull. Int. Dairy Fed., 375: 59-64.
Direct Link - Okhuysen, P.C., C.L. Chappell, J. Crabb, L.M. Valdez, E.T. Douglass and H.L. DuPont, 1998. Prophylactic effect of bovine anti-Cryptosporidium hyperimmune colostrums immunoglobulin in healthy volunteers challenged with Cryptosporidium parvum. Clin. Infect. Dis., 26: 1324-1329.
Direct Link - Oona, M., T. Rago, H.I. Maaroos, M. Mikelsaar, K. Loivukene, S. Salminen and H. Korhonen, 1997. Helicobacter pylori in children with abdominal complaints: Has immune bovine colostrum some influence on gastritis? Alpe Adria Microbiol. J., 6: 49-57.
Direct Link - Pakkanen, R. and J. Aalto, 1997. Growth factors and antimicrobial factors of bovine colostrum. Int. Dairy J., 7: 285-297.
Direct Link - Reilly, R.M., R. Domingo and J. Sandhu, 1997. Oral delivery of antibodies. Clin. Pharmacokient, 32: 313-323.
PubMedDirect Link - Roos, N., S. Mahe, R. Benamouzig, H. Sick, J. Rautureau and D. Tome, 1995. 15-N-labeled immunoglobulins from bovine colostrums are partially resistant to digestion in human intestine. J. Nutr., 125: 1238-1244.
PubMedDirect Link - Sarker, S.A., T.H. Casswall, D. Mahalanabis, N.H. Alam and M.J. Albert et al., 1998. Successful treatment of rotavirus diarrhea in children with immunoglobulin from immunized bovine colostrum. Pediatr. Infect. Dis. J., 17: 1149-1154.
PubMedDirect Link - Virtanen, S., E.L. Syvaoja, V. Loimaranta and H. Korhonen, 1998. Stability of activity of bovine colostrum derived anti-caries streptococci antibodies added to a UHT milk product. Proceedings of the European Dairy Experts Symposium: Dairying behind the Dikes, September 15-18, 1998, Arnhem, The Netherlands, pp: 38.
- Warny, M., A. Fatimi, E.F. Bostwick, D.C. Laine, J.T. LaMont, C. Pothoulakis and C.P. Kelly, 1999. Bovine immunoglobulin concentrate-Clostridium difficile retains C difficile toxin neutralizing activity after passage through the stomach and small intestine. Gut, 44: 212-217.
Direct Link - Weiner, C., Q. Pan, M. Hurtig, T. Boren, E. Bostwick and L. Hammarstrm, 1999. Passive immunity against human pathogens using bovine antibodies. Clin. Exp. Immunol., 116: 193-205.
CrossRefPubMedDirect Link - Wie, H., P. Marnila and H. Korhonen, 2002. Effects of anti-caries antibodies on Lactobacillus GG in its fermentation and storage periods. Biomed. Environ. Sci., 15: 153-165.
PubMedDirect Link








