Research Article
Single Cell Protein: Production and Process
Department of Pharmaceutical Biotechnology and Pharmaceutical,Sciences Research Center, Faculty of Pharmacy,Shiraz University of Medical Sciences, P.O. Box 71345-1583, Shiraz, Iran
S. Rasoul-Amini
Department of Pharmaceutical Biotechnology and Pharmaceutical,Sciences Research Center, Faculty of Pharmacy,Shiraz University of Medical Sciences, P.O. Box 71345-1583, Shiraz, Iran
M.H. Morowvat
Department of Pharmaceutical Biotechnology and Pharmaceutical,Sciences Research Center, Faculty of Pharmacy,Shiraz University of Medical Sciences, P.O. Box 71345-1583, Shiraz, Iran
LiveDNA: 98.36188
Y. Ghasemi
Department of Pharmaceutical Biotechnology and Pharmaceutical,Sciences Research Center, Faculty of Pharmacy,Shiraz University of Medical Sciences, P.O. Box 71345-1583, Shiraz, Iran










Md. Aabid Hussain Reply
Great !! Good job....very simple language and easy to understand...
Thank You.
Regards:
Aabid
Achmad Farits Fauzi Reply
Subhanalloh ...
Thanks sir ...
I hope this journal can use for next time ...
Dr.sushil mathur Reply
I would like a copy of this article.kindly mail thanks in anticipation,& hope a positive reply.Mathur
uduakobong raphael akpan Reply
It is a good work.pls can send me biotechnology aspect of scp production.thanks
shahab uddin Reply
comprehensive and easy to get.... shahab
Jane S. Fom Reply
I love the work and would like to have a copy.
Asma BEN TEKAYA Reply
awesome
Mayang Reply
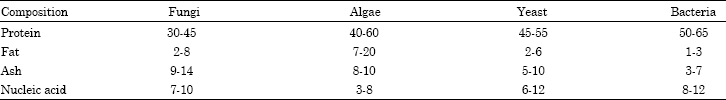
The concept of producing protein from microorganisms like algae, fungi, yeast, and bacteria is an innovative approach to address the worldwide protein shortage. The use of inexpensive feedstock and wastes as sources of carbon and energy for growth to produce biomass or protein concentrate is an added advantage. Single cell proteins have high nutritive value with higher protein, vitamin, essential amino acids, and lipid content. However, the concern over their high nucleic acid content, slower digestibility, and potential for allergic reactions may raise questions about their widespread use as a replacement for conventional protein sources. Further research and development in this area could address these concerns and contribute to sustainable protein production.