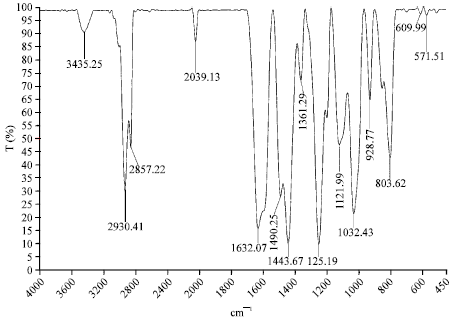
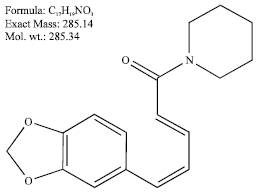
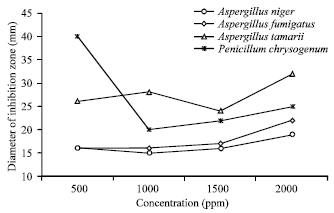
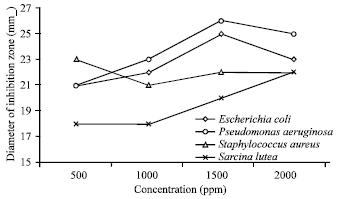
Research Article
Antioxidant and Antimicrobial Activities of the Hot Pungent Chabbarin are Responsible for the Medicinal Properties of Piper chaba, Hunter
Agricultural and Ecological Research Unit, Indian Statistical Institute, 203 B.T. Road, Calcutta 700 108, India
Nabanita Chakraborty
Agricultural and Ecological Research Unit, Indian Statistical Institute, 203 B.T. Road, Calcutta 700 108, India
Priyanka Chakraborty
Departmant of Microbiology, 3Department of Biotechnology, University of Kalyani, Kalyani 741234, Nadia, India
Sampad Sarkar
Departmant of Microbiology, 3Department of Biotechnology, University of Kalyani, Kalyani 741234, Nadia, India