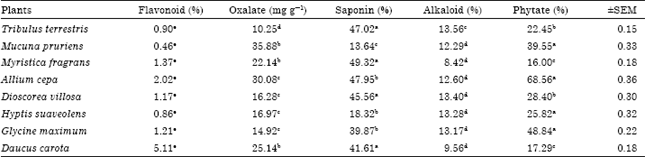
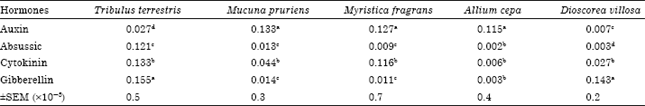
Research Article
Investigation of Phytohormonal Potential of Some Selected Tropical Plants
Reproductive Physiology Unit, Department of Animal Production, Faculty of Agriculture, University of Ilorin, P.M.B. 1515, Ilorin, Kwara State, Nigeria
A.A. Adeloye
Reproductive Physiology Unit, Department of Animal Production, Faculty of Agriculture, University of Ilorin, P.M.B. 1515, Ilorin, Kwara State, Nigeria
M.A. Belewu
Reproductive Physiology Unit, Department of Animal Production, Faculty of Agriculture, University of Ilorin, P.M.B. 1515, Ilorin, Kwara State, Nigeria
O.I. Alli
Reproductive Physiology Unit, Department of Animal Production, Faculty of Agriculture, University of Ilorin, P.M.B. 1515, Ilorin, Kwara State, Nigeria
O.A. Adeyina
Reproductive Physiology Unit, Department of Animal Production, Faculty of Agriculture, University of Ilorin, P.M.B. 1515, Ilorin, Kwara State, Nigeria
A.A. Annongu
Reproductive Physiology Unit, Department of Animal Production, Faculty of Agriculture, University of Ilorin, P.M.B. 1515, Ilorin, Kwara State, Nigeria