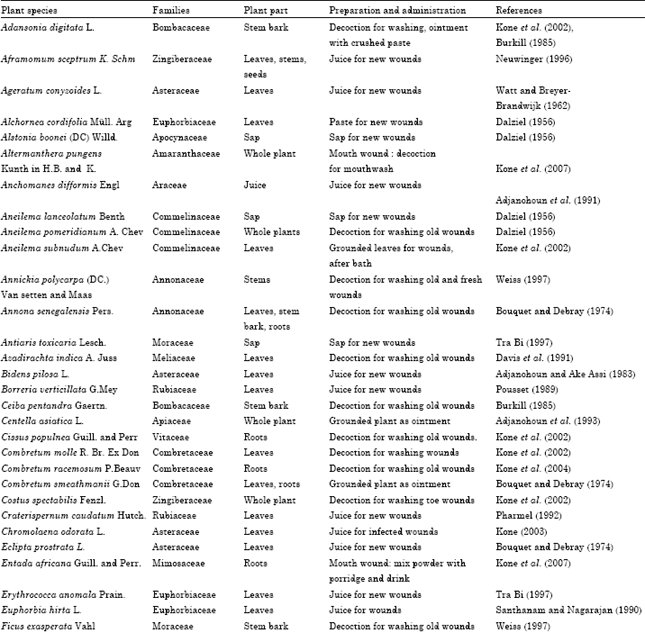
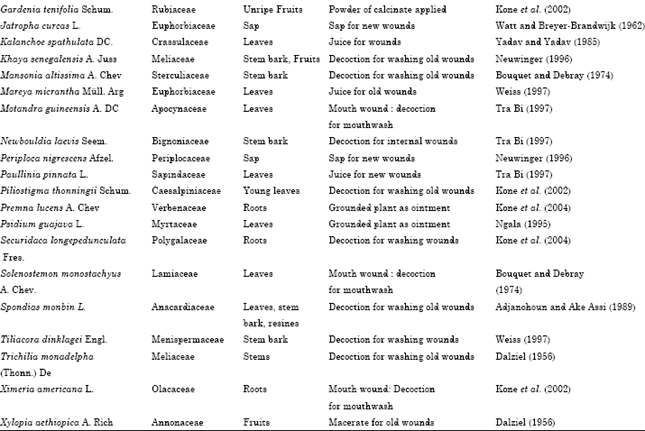
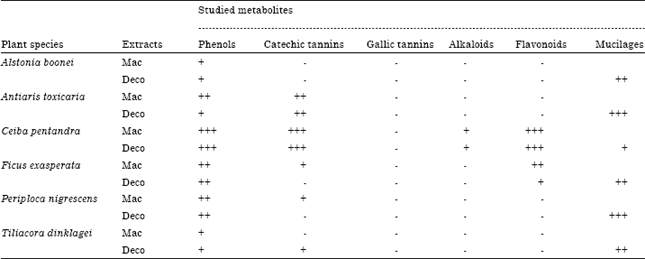
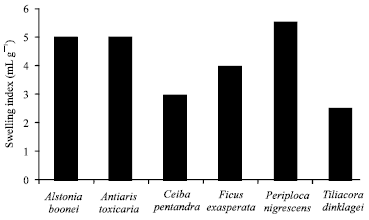
Research Article
Preliminary Study on Polysaccharides and Certain Secondary Metabolites of Medicinal Plants used in Cote D’Ivoire for Wound Healing
UFR Sciences de la Nature, Universite d`Abobo-Adjame, 02 B.P. 801 Abidjan 02, Cote d�Ivoire
A. Azokou
UFR Sciences de la Nature, Universite d`Abobo-Adjame, 02 B.P. 801 Abidjan 02, Cote d�Ivoire
A. Bakayoko
UFR Sciences de la Nature, Universite d`Abobo-Adjame, 02 B.P. 801 Abidjan 02, Cote d�Ivoire
F.H. Tra Bi
UFR Sciences de la Nature, Universite d`Abobo-Adjame, 02 B.P. 801 Abidjan 02, Cote d�Ivoire