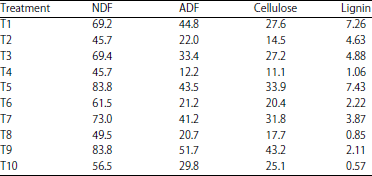
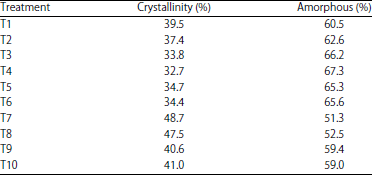
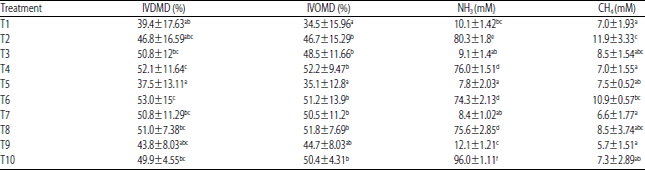
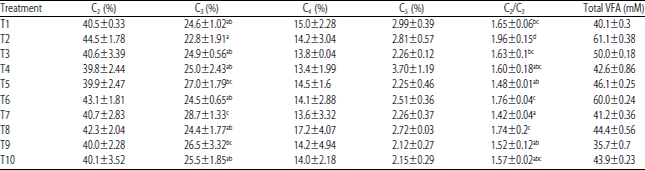
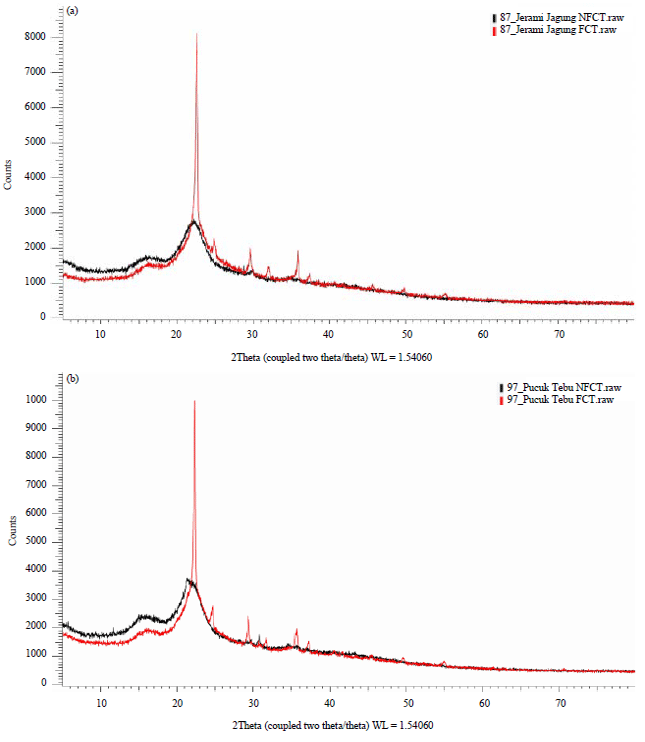
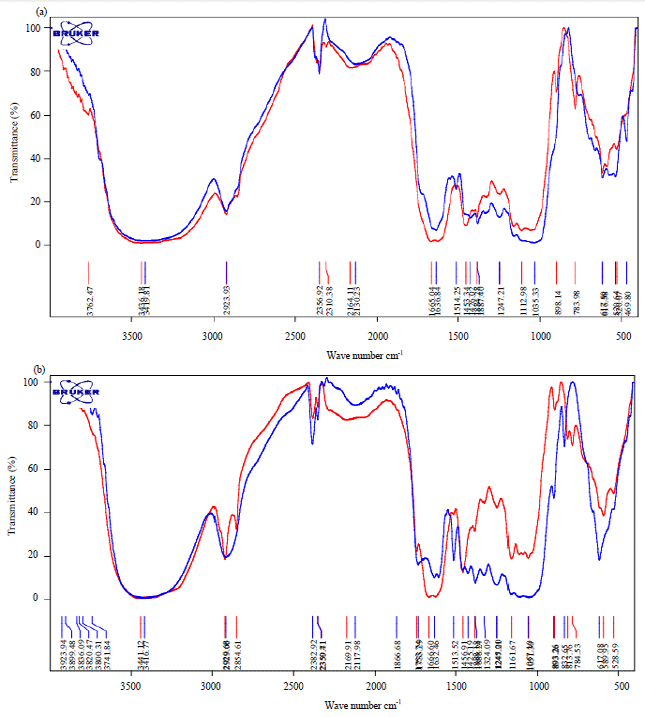
Research Article
Use of Fiber Cracking Technology to Improve the Nutritive Quality of Corn and Sugarcane By-products for Ruminant Feeds
Department of Animal Nutrition and Feed Technology, Faculty of Animal Science, Bogor Agricultural University, 16680 Bogor, Indonesia
Muhammad Ridla
Department of Animal Nutrition and Feed Technology, Faculty of Animal Science, Bogor Agricultural University, 16680 Bogor, Indonesia
Erika B. Laconi
Department of Animal Nutrition and Feed Technology, Faculty of Animal Science, Bogor Agricultural University, 16680 Bogor, Indonesia
Anuraga Jayanegara
Department of Animal Nutrition and Feed Technology, Faculty of Animal Science, Bogor Agricultural University, 16680 Bogor, Indonesia
LiveDNA: 62.7299