Research Article
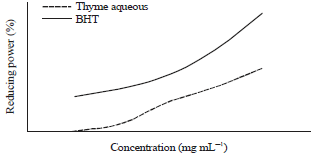
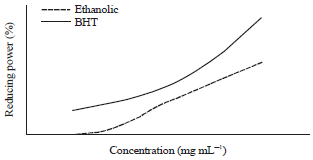
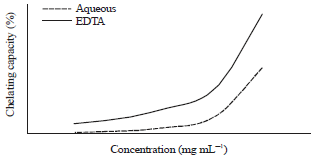
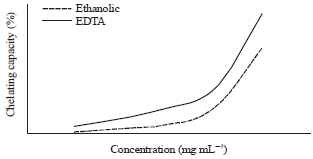
Antioxidant Activities of Thyme Extracts
Basic Science Section, College of Agriculture, University of Baghdad, Baghdad, Iraq
T.K. Nahla
Department of Food Science, College of Agriculture, University of Baghdad, Baghdad, Iraq
LiveDNA: 964.19844
N.M. Tariq
Department of Food Science, College of Agriculture, University of Baghdad, Baghdad, Iraq