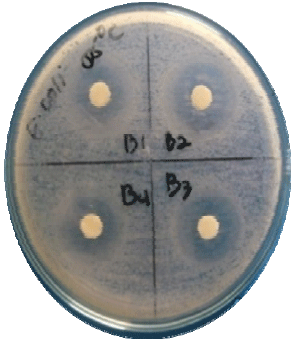
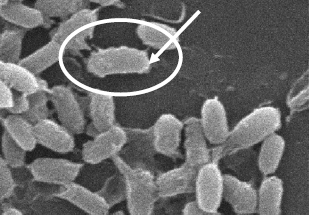
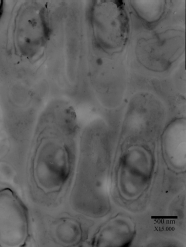
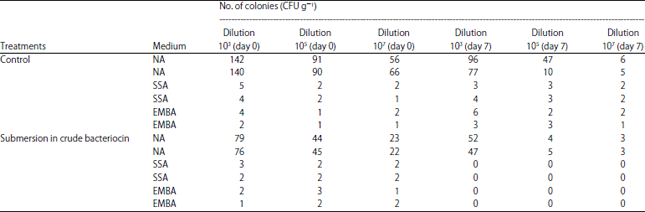
Research Article
Effect of the Bacteriocin-producing Bacillus cereus Strain HVR22 on the Preservation of Fish Fillets
Department of Fisheries and Marine Sciences, Bung Hatta University, 25133 Padang, Indonesia
Yempita Efendi
Department of Fisheries and Marine Sciences, Bung Hatta University, 25133 Padang, Indonesia