Research Article
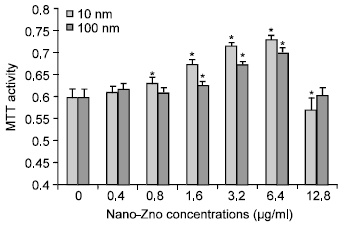
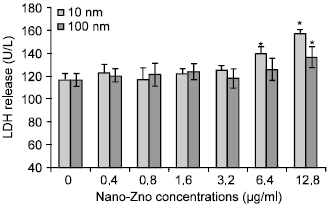
The Effects of Different Sizes of Nanometer Zinc Oxide on the Proliferation and Cell Integrity of Mice Duodenum-Epithelial Cells in Primary Culture
Institute of Animal Nutrition, Sichuan Agricultural University, Ya�an, Sichuan 625014, China
Z. S. Wang
Institute of Animal Nutrition, Sichuan Agricultural University, Ya�an, Sichuan 625014, China
A. G. Zhou
Key Laboratory for Animal Disease, Resistance Nutrition of China, Ministry of Education, Ya�an, Sichuan 625014, P.R. China
D. W. Ai
Key Laboratory for Animal Disease, Resistance Nutrition of China, Ministry of Education, Ya�an, Sichuan 625014, P.R. China