Research Article
Molecular Diagnosis of Naturally Infection with Eimeria nieschulzi in Laboratory Rats
Department of Veterinary Parasitology, College of Veterinary Sciences and A.H., Central Agricultural University, Aizawl, Mizoram, India
M. Ayub Ali
Department of Veterinary Biochemistry and Physiology, College of Veterinary Sciences and A.H., Central Agricultural University, Aizawl, Mizoram, India
Kh. Victoria Chanu
Department of Veterinary Biochemistry and Physiology, College of Veterinary Sciences and A.H., Central Agricultural University, Aizawl, Mizoram, India
Jonathan Lalsiamthara
SRF-RCRL, College of Veterinary Sciences and A.H., Central Agricultural University, Aizawl, Mizoram, India
J.L. Kataria
SRF-RCRL, College of Veterinary Sciences and A.H., Central Agricultural University, Aizawl, Mizoram, India
Snigdha Hazarika
Department of Veterinary Pharmacology and Toxicology, College of Veterinary Sciences and A.H., Central Agricultural University, Aizawl, Mizoram, India
David Malsawmkima
Department of Veterinary Microbiology, College of Veterinary Sciences and A.H., Central Agricultural University, Aizawl, Mizoram, India
R. Ravindran
Department of Veterinary Pathology, College of Veterinary Sciences and A.H., Central Agricultural University, Aizawl, Mizoram, India
L. Inaotombi Devi
Department of Medical Laboratory Technology, RIPANS, Aizawl, Mizoram, India









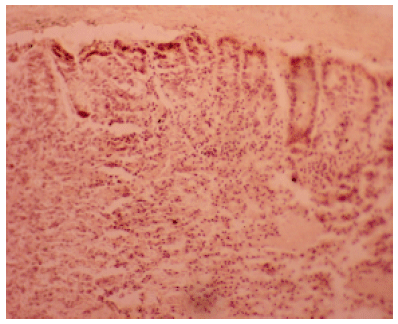
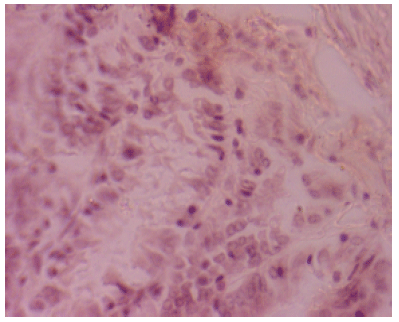
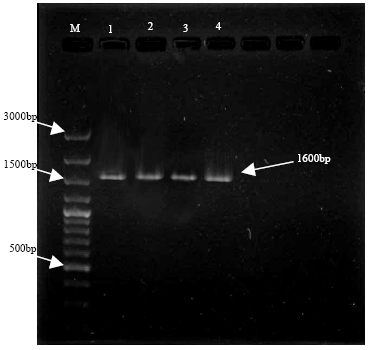
Dr. Michael Kurth Reply
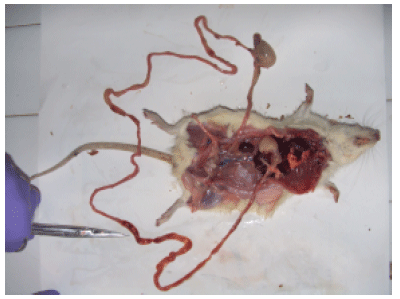
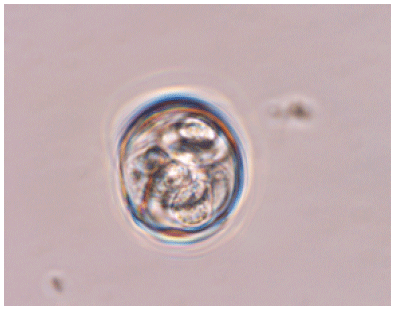
I´ve some doubts that the identified species is Eimeria nieschulzi. We work for a long time with Eimeria nieschulzi and we never saw such necrotic tissue in the small intestine and never in the large intestine. Lesions caused by Eimeria nieschulzi are normally limited on the small intestine. Further, the shown oocyst in Figure 3 is (at least) not a (typical) Eimeria nieschulzi oocyst. The histological images have unfortunatly a too poor quality to see if the developmental stages are in the villi or in the crypts. With the used primer pair it is not possible to amplify the specific 18S rDNA of Eimeria nieschulzi. By the used primers you can amplify the 18S rDNA of every Eimeria species.The PCR reaction wasn´t a proof without sequencing.