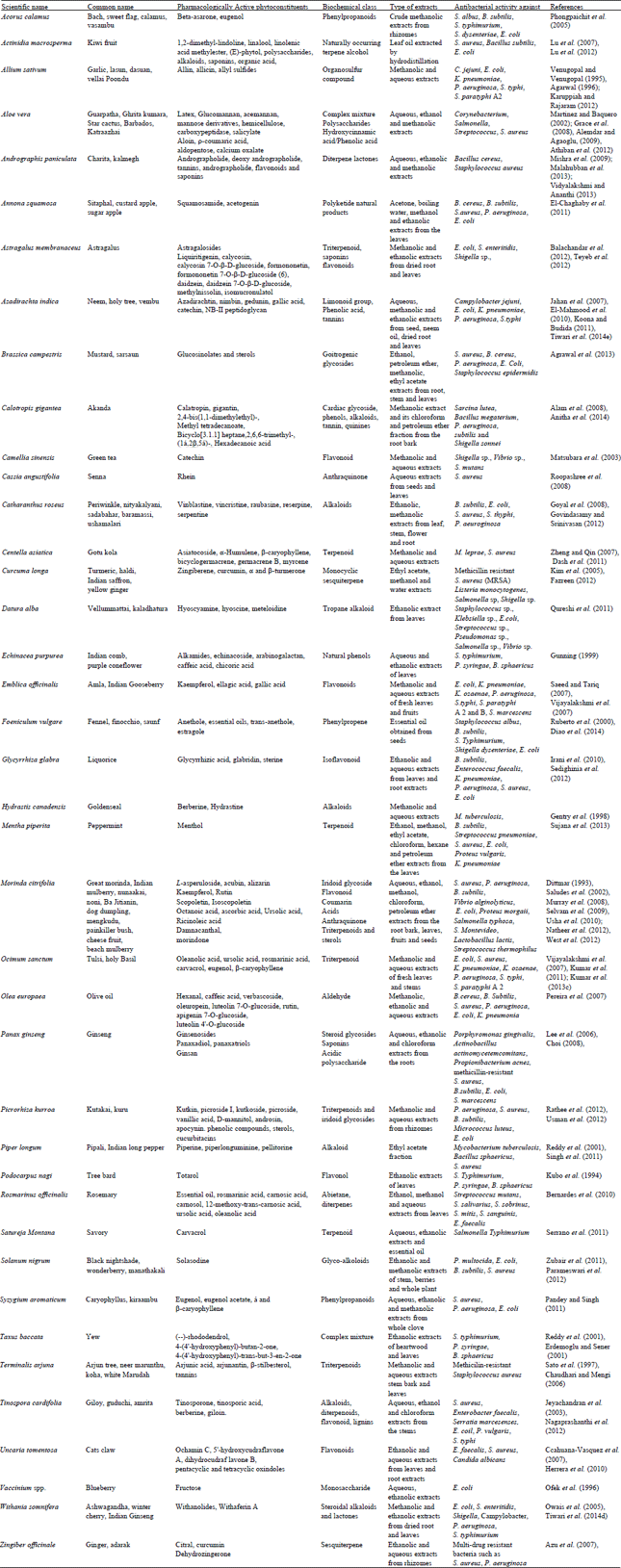
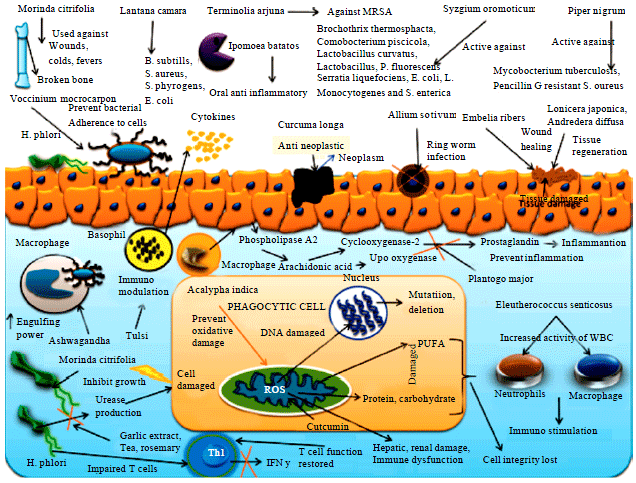
Review Article
Evidence Based Antibacterial Potentials of Medicinal Plants and Herbs Countering Bacterial Pathogens Especially in the Era of Emerging Drug Resistance: An Integrated Update
Department of Pathology,Pt. Nehru Complex, Agartala, 799006, Pin, India
Ruchi Tiwari
Department of Veterinary Microbiology and Immunology,Pt. Nehru Complex, Agartala, 799006, Pin, India
Sandip Chakraborty
Department of Animal Resources Development, Pt. Nehru Complex, Agartala, 799006, Pin, India
Mani Saminathan
Department of Pathology,Pt. Nehru Complex, Agartala, 799006, Pin, India
Amit Kumar
Department of Veterinary Microbiology and Immunology,Pt. Nehru Complex, Agartala, 799006, Pin, India
K. Karthik
Department of Bacteriology and Mycology,Khalsa College of Veterinary and Animal Sciences (KCVAS), Amritsar, 143001, Punjab, India
Mohd. Yaqoob Wani
Department of Veterinary Microbiology, Khalsa College of Veterinary and Animal Sciences (KCVAS), Amritsar, 143001, Punjab, India
Amarpal
Department of Surgery, Indian Veterinary Research Institute, Izatnagar (IVRI),Bareilly, 243122, Uttar Pradesh, India
Shoor Vir Singh
Department of Microbiology Laboratory, Animal Health Division,Central Institute for Research on Goats (CIRG), Makhdoom,PO-Farah, District Mathura, 281122, Uttar Pradesh, India
Anu Rahal
Department of Veterinary Pharmacology and Toxicology, College of Veterinary Sciences and Animal Husbandry, Uttar Pradesh Pandit Deen Dayal Upadhayay Pashu Chikitsa Vigyan Vishvidhyalaya Ewam Go-Anusandhan Sansthan (DUVASU), Mathura, 281001, Uttar Pradesh, India