Review Article
Diagnosis and Control of Foot and Mouth Disease (FMD) in Dairy Small Ruminants; Sheep and Goats
Department of Parasitology and Animal Diseases, Veterinary Research Division, National Research Centre, Dokki, Cairo, Egypt
LiveDNA: 20.20509
Alaa Abdelmoneam Ghazy
Department of Parasitology and Animal Diseases, Veterinary Research Division, National Research Centre, Dokki, Cairo, Egypt
LiveDNA: 20.1024
Raafat Mohamed Shaapan
Department of Zoonotic Diseases, Veterinary Research Division, National Research Centre, Dokki, Giza, Egypt
LiveDNA: 20.659









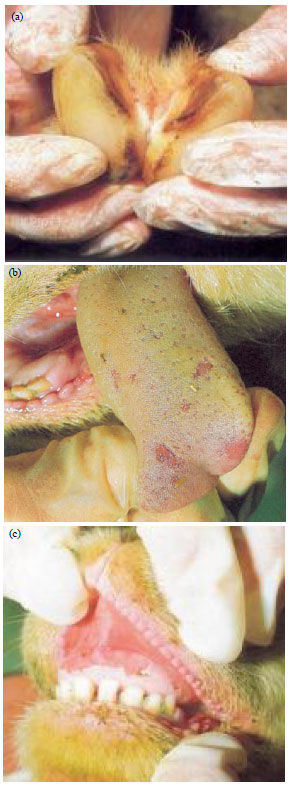
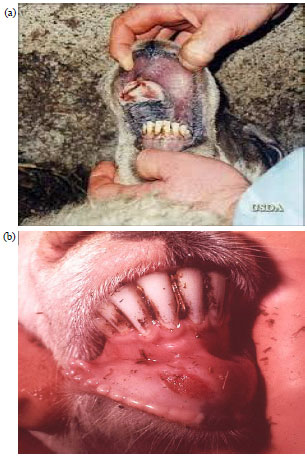
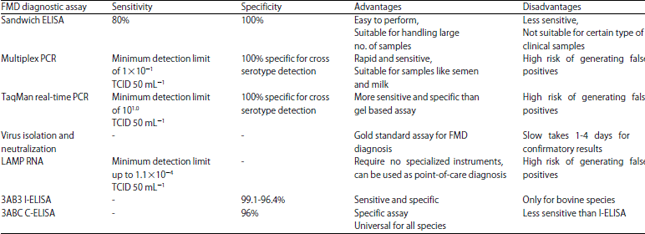
Elachi Johnbona S. A. (DVM) Reply
thank you for the good job! keeping veterinary profession high. I HAVE A PAPER TO PUBLISH BUT IT IS ON WISTAR RAT!