ABSTRACT
Two experiments were carried out to evaluate the effects of laboratory produced tannase enzyme (Tanozym) to diet including Palm Kernel Powder (PKP) on in vitro dry matter and organic matter disappearance (IVDMD and IVOMD) and in vivo nutrients digestibility, nutritive values, milk production and composition by lactating Baladi goats. In vitro experiment, IVDMD and IVOMD, were determined for control diets (60% CFM and 40% Berseem hay); (T1) control diet plus different levels of polyethylene glycol (PEG), being 10, 15 and 20 g kg-1 DM and (T2) control diet plus different levels of Tanozym (3.9, 5.85 and 7.8 U kg-1 DM). The maximum IVDMD and IVOMD values were observed with 5.85 U kg-1 DM for Tanozym and 20 g kg-1 DM for PEG compared to control, however there was no significant (p<0.05) difference between 15 and 20 g kg-1 DM. The in vivo experiment was carried out on nine lactating Baladi goats after 7 days of parturition where animals were divided into three groups, three animals each, using 3x3 Latin square design. The first group fed control diet (60% CFM and 40% Berseem hay), the second group fed T1 (control diet plus 15 g kg-1, DM), the third group fed T2 (control diet plus 5.85 U kg-1, DM). Tanozym supplementation significantly (p<0.05) increased nutrients digestibility, nutritive values, ruminal Total Volatile Fatty Acids (TVFA’s) but insignificant (p<0.05) increased ammonia nitrogen (NH3 N). Lower significant (p<0.05) values of rumen pH were recorded for treated groups compared with the control. Blood serum of animals fed Tanozym and PEG had higher values of total protein, albumin, globulin, total lipids, urea and glucose but lower values of AST and ALT compared with those of control. Daily milk yield, SNF, lactose and ash yield were significantly (p<0.05) increased with Tanozym compared control group. While, there are no significant (p>0.05) differences among groups for fat corrected milk 4%, total solids, fat and total protein yield.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/ijds.2015.24.35
URL: https://scialert.net/abstract/?doi=ijds.2015.24.35
INTRODUCTION
Anti-Nutritional Factors (ANFs) are natural or synthetic substances which, when present in human foods and animals feed, reduce growth and interfere with the absorption of nutrients (Hotz and Gibson, 2007). Examples of these ANFs are tannins, phytate, protease and excessive dietary fiber. Tannin compounds are widely distributed in many species of plants, where they play a role in protection from predation and perhaps as pesticides and in plant growth regulation (Katie and Thorington, 2006). The problems of feeding agricultural by-products to farm animals are regarded to its low protein content, high crude fiber, low nutrients digestibility coefficients and its high content of anti-nutrients factors such as tannins and alkaloids (Kholif et al., 2005; Khattab et al., 2008). Addition of polyethylene glycol had beneficial effects in monogastrics and both beneficial and adverse effects in ruminants depend upon amount of tannins in the diet (Silanikove et al., 1994, 1996). Polyethylene glycol is a nonionic detergent which forms complexes with hydrolysable and condensed tannins over a wide pH range (2-8.5) (Jones, 1965). Protein may be released from the protein-tannin complex by exchange reaction with PEG (Jones and Mangan, 1977). Tannase enzyme can hydrolyzes tannins substrates such as methyl gallate, propyl gallate, digallic acid, epicatechin gallate and epigallocatechin gallate-releasing gallic acid (Curiel et al., 2009; Lu and Chen, 2007). The main objective of this study was to determine the effect of using laboratory produced tannase enzyme (Tanozym) for reducing negative tannins effect (as an anti-nutritional factor) on nutrients digestion coefficient, nutritive values, rumen and blood parameters and milk production and composition of lactating Baladi goats.
MATERIALS AND METHODS
The present experiment was carried out at a private farm in Atfih city, Helwan, Egypt and at the Department of Dairy Science, National Research Center, Dokki, Giza, Egypt.
Enzyme sources: Laboratory produced tannase enzyme from Asperigillus niger. Each killogram contained 780 international unit of tannase was used.
Assay of tannase: Tannase enzyme activity was determined by the method of Mondal et al. (2001). One unit of the tannase enzyme was defined as the amount of enzyme which is able to hydrolyse 1 μ mole of ester linkage of tannic acid in 1 min at specific condition (pH 5.0 and 40°C).
In vitro study: Three experimental diets were used to investigate the effect of in vitro DM and OM disappearance (IVDMD and IVOMD). The experimental diets were; control diet was total mixed ration consist of 60% Concentrate Feed Mixture (CFM) and 40% roughage (Berseem hay), the chemical composition of CFM and Berseem hay were showed in Table 1; (T1) control plus different levels from Tanozym (3.9, 5.85 and 7.8 U kg-1 DM); (T2) control plus different levels from Poly Ethylene Glycol (PEG), (MW 20000) (10, 15 and 20 g kg-1 DM).
| Table 1: | Chemical composition of feed ingredients (on DM basis) |
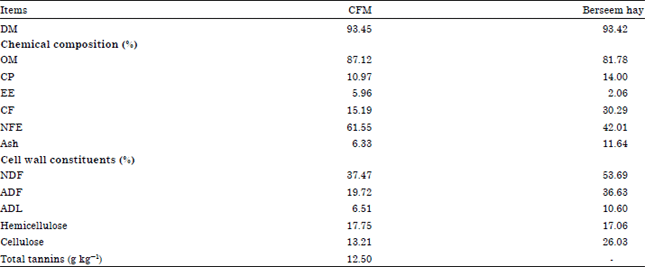 | |
| Hemicellulose: NDF-ADF, Cellulose: ADF-ADL and CFM: Concentrate feed mixture | |
Thirty five incubation flask (250 mL volume) were used (5 flasks per each treatments) used to determine (IVDMD and IVOMD). The procedures of the in-vitro technique were carried out according to Fondevila and Perez-Espes (2008). The procedures were using flasks filled with 140 mL of incubation solution, (Theodorou et al., 1994) prepared under a CO2 atmosphere, including a buffer solution, macro-mineral and trace mineral solution, a reduction solution and rumen inoculum. Rumen liquor were collected from goats fed berseem hay ration before the morning feeding by using stomach tube, then moved directly to the laboratory in separate warmed oxygen-free thermos flask. Rumen liquor contents were strained through two layers of cheese-cloth. Flasks were sealed with rubber stoppers and incubated at 39°C in a shaking water bath (20 oscillations min-1) for 48 h.
Lactation experiment: According to results of in vitro experiment, the proper IVDMD and IVOMD of different levels of Tanozym and PEG, Tanozym at 5.85 U kg-1 DM and PEG at 15 g kg-1 DM were chosen to be used in the location experiment.
Animals and diets: Nine lactating Baladi goats of about 2-4 years age and averaged about 23.5 kg live body weight after 7 days of parturition were divided randomly (three groups of three animals each) using 3x3 Latin square design. The experimental periods were extended for 12 weeks (84 days) and consisted of three equal periods (28 day of each). Each period involved 21 days as preliminary period followed by 7 days for collection of the experimental samples. Goats were individually fed amounts equal 4% of live body weight on experimental diets which consisted of 60:40 concentrate:roughage ratio (on DM basis). The CFM consisted of 25% maize, 25% PKP, 20% cotton seed meal, 15% wheat bran, 12% barley grain and 1% limestone, 1% NaCl and 1% minerals-vitamins premix. The experimental diets Control diet: 60% CFM and 40% berseem hay, T1: Control diet plus PEG at 15 g kg-1 and T2: Control diet plus Tanozym at 5.85 U kg-1. The CFM was offered twice daily at 6 a.m. and at 6 p.m. then berseem hay was offered twice daily at 7 a.m. and 7 p.m. The enzyme and PEG were introduced to each animal as a mixer with CFM. Dry matter intake was measured during the last 7 days of experimental period. The fresh water was always available to animals.
Analysis of feed and fecal samples: Samples of ingredients and rations were analyzed for Dry Matter (DM), ash, Crude Fiber (CF), Organic Matter (OM) and Ether Extract (EE) according to method of AOAC (1995) while, Nitrogen-Free Extract (NFE) was calculated by difference. The Neutral Detergent Fiber (NDF) and Acid Detergent Fiber (ADF) contents were determined using the methods described by Van Soest (1994). Total tannins were determined according to Hagerman and Butler (1978).
Determination of digestion coefficients: Grab sample method was applied for determining the nutritive value and digestibility in which Acid Insoluble Ash (AIA) was used as an internal marker (Gallup et al., 1945; Forbes and Garrigus, 1948). Feces grab samples were collected manually at 5 a.m. for 3 successive days at the first 3 days of collection periods for each experiment. Representative feces samples were sprayed by 10% sulfuric acid solution and formalin, then dried in oven at 70°C for 24 h. The fresh feces samples for each animal were mixed well by equal weights then ground and stored in polyethylene bags for chemical analysis of DM, OM, CP, CF, NDF, ADF and ash. The digestibility coefficient of a certain nutrient was calculated according to the following equation:
![]()
Sampling and analysis of rumen liquor: Rumen liquor samples were collected by stomach tube at the last day of each experimental period for each animal. Samples were taken before morning feeding (zero time) and at 3 and 6 h after morning feeding. The rumen samples were filtered through two layers of cheese cloth and were immediately used for determination of ruminal pH by digital pH-meter, then rumen liquor samples were stored in glass bottles with drops of toluene and thin layer of paraffin oil in a deep freezer at -18°C for analysis. Ruminal pH was determined using a digital pH meter and NH3 N were determined (AOAC, 1995). Total Volatile Fatty Acids (TVFA’s) were determined by steam distillation as described by Warner (1964).
Sampling and analysis of blood serum: Blood samples were taken from each animal at the last day of each experimental period. The samples (10 mL of blood per animal) were taken before morning feeding (zero time) and at 4 h post feeding. Each blood sample was withdrawn from the jugular vein into a clean dried tube and centrifuged at 4000 rpm for 15 min, then blood serum was separated into a clean dried glass vial and stored in deep freezer at -18°C for analysis. Serum total protein was determined (Gornal et al., 1949), serum albumin (Doumas et al., 1971), serum globulin and A:G ratio were calculated, serum aspartate aminotransferase (AST) and alanin aminotransferase (ALT) (Reitman and Frankel, 1957), serum urea (Fawcett and Scott, 1960), serum glucose (Trinder, 1969) and serum total lipids (Zollner and Kirsch, 1962).
Sampling and analysis of milk: The animals were hand milked twice daily at 5 a.m. and 5 p.m. during the last 3 days of each experimental period. Samples of milk were collected from each animal at morning and evening milking immediately. The representative sample of each animal was a mixed sample of constant percentage of the evening and morning yield for total chemical analysis. Milk samples were analyzed for total solids, solids not fat, total protein, fat, ash and lactose using infrared spectroscopy (Bentley 150, Infrared Milk Analyzer, Bentley Instruments, USA). Fat corrected milk (4% fat) was calculated by using the following equation (Gaines, 1927):
FCM = 0.4 M+15 F
Where:| M | = | Milk yield (g d-1) |
| F | = | Fat yield (Amount of fat = M×fat %) |
Statistical analysis: The data was analyzed using general linear method of statistical analysis system (SAS, 1998) and Duncan multiple range test (Duncan, 1955) was carried out for separation among means:
| • | Data of milk yield, milk composition and nutrients digestibility was analyzed according to Latin square design where the model was: |
Yijk = μ+RI+Cj+Tk+eijk
where, Yijkl is the parameter under analysis of the ijkl goat, μ is the overall mean, Ri is the effect due to the lactation period on the parameter under analysis, Cj is the effect due to the animals on the parameter under analysis, Tk is the effect due to treatment on the parameter under analysis, eijk is the experimental error for ijk on the observation
| • | Repeated measures for rumen liquid parameter and blood serum parameters were described as: |
Yijk = μ+RI+Tj+(RT)ij+Bk+(TB)jk+(RB)ik+Eijk
where, RI is period, Tj is treatment, (RT)ij is interaction, Bkt is sampling time, (TB)jk is interaction (TB)jk, (RB)ik is interaction and Eijk is experimental error.
RESULTS AND DISCUSSION
In vitro study: Table 2 showed the effect of Tanozym and PEG supplementations on IVDMD and IVOMD. Results indicated that all levels of PEG (T1) and Tanozym (T2) supplementation were associated with increase of IVDMD and IVOMD values for diets compared to Control. Increasing the supplementation levels up to 5.85 U kg-1 DM gave significant (p<0.05) increase of both (IVDMD and IVOMD) but the highest level (7.8 U kg-1 DM) decreased with the Tanozym. While, PEG gave the highest IVDMD and IVOMD values at 20 g kg-1 DM but there was no significant (p<0.05) increase between PEG at 15 and 20 g kg-1 DM. Improvement of IVDMD and IVOMD could be attributed to the influence of tannase and PEG on tannin reduction similar results were reported by Frutos et al. (2004) and Lekha and Lonsane (1997). The present results were accepted by Angaji et al. (2011) who found that adding PEG increased IVOMD and IVDMD.
This result may also be related to type of tannins and bacterial species in the rumen inoculum, factors which influence the ability of PEG to inhibit effects of tannins (Frutos et al., 2004; Angaji et al., 2011). The use of tannase as an ingredient of animal feed would improve the digestibility of the feed (Lekha and Lonsane, 1997). Dong et al. (1999) illustrated that the effects of enzymes might start when the enzyme is in contact with the substrate, so enzyme-feed interaction appears as important. Giraldo et al. (2004) confirmed that a pre-ingestive enzyme-feed interaction is necessary for any significant beneficial effects on ruminal digestion.
| Table 2: | Effect of treated with tanozym and PEG on in vitro dry matter and organic matter disappearance |
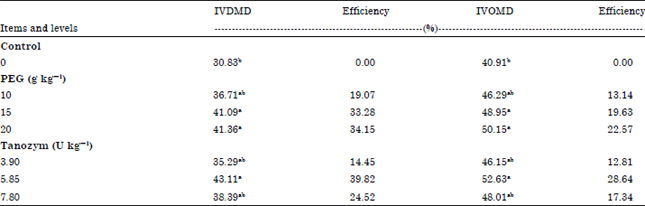 | |
| a,b,cMeans with different superscripts are significant (p<0.05). Efficiency (%) (DM) = IVDMD (%) (sample)-IVDMD (%) (control)/IVDMD (%) (control)x100, Efficiency (%) (OM) = IVOMD (%) (sample)-IVOMD (%) (control)/IVOMD (%) (control)x100, Means designated with the same letter in the same column are not significantly different at 0.05 level of probability | |
| Table 3: | Digestibility co-efficient of nutrients, nutritive value of experimental diets fed to goats |
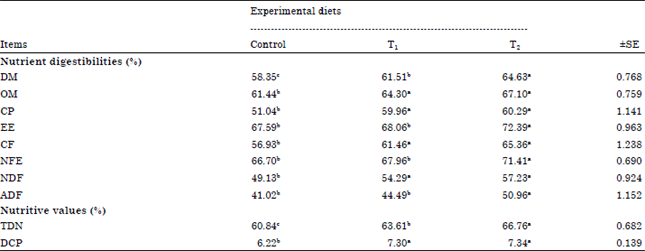 | |
| a,b,cMeans with different superscripts are significant (p<0.05), SE: Standard error | |
The enzyme addition onto feeds may create a stable enzyme-feed complex that protects free enzymes from proteolysis in the rumen as reported by Kung et al. (2000).
Digestibility of nutrients and nutritive value: Table 3 showed that there were significant (p<0.05) increase in the digestibilities of OM, CP, CF and NDF between T1 and T2 compared with control. But the results were insignificant (p>0.05) differences in these nutrients digestibilities between T1 and T2. Also, there were significant (p<0.05) increase in the digestibilities of DM, EE, NFE and ADF between T2 compared with Control and T1. While, the results were insignificant (p>0.05) differences in these nutrients digestibilities between control and T1. Enhancing in nutrients digestibilities attributed to reduce effect of tannins content by treated with Tanozym and PEG. These results were accepted by Khattab et al. (2008) who found that there was significant (p<0.05) increase of all nutrients digestibilities (DM, OM, CP, CF and NFE), that may be attributed to the reduction of tannins and alkaloids in the by-products silage. It was reported by Makkar (2003) that tannins reduce degradability of protein and other nutrients. The present results are also in a line with those reported by Abdel-Halim (2003) who found that ensiling process reduced tannins in the ration from 12.6-8.8%, hence the DM digestibility improved from 48.4-58.5% this negative influence of phenolics on digestibility has been attributed to their toxic effects on rumen microbes (Tolera et al., 1997). Enhancing in T1 attributed to PEG makes tannins inert by forming tannin-PEG complexes (Makkar et al., 1995). There was significant (p<0.05) increase in the nutritive values as TDN in T2 compared with other groups. Also, there were significant (p<0.05) increase in the nutritive values as DCP between T1 and T2 compared with control. These results are in agreement with those obtained by Khattab et al. (2008) who found that increased TDN and DCP with reduce tannins and alkaloids contents with silage. While, Hassan (2006) found the same results when feeding animals on rations containing tannins treated with PEG. However, these results disagree with Barry et al. (1986) who found that decreases of TDN and DCP treatment of PEG are due to the protein liberating effect of PEG from the tannin-protein complexes.
Rumen liquor parameters: Ruminal pH showed significant (p<0.05) higher values by goats fed control diets compared with (T2) diet (Table 4) while, no significant (p>0.05) differences between (control and T1) and (T1 and T2).
| Table 4: | Effect of experimental diets on some rumen liquor parameters |
 | |
| a,b,cMeans with different superscripts are significant (p<0.05), SE: Standard error | |
These result caused by the intensive fermentation process of both non structural and structural carbohydrates and the production of volatile fatty acids. Such results are supported by the finding of Yanez Ruiz et al. (2004) who fond that adding PEG supply seemed to stimulate carbohydrate fermentation in goats and wethers because it promoted a pH decrease. The increases of pH with the control diet means that the tannins reduce the activity of bacterial proteolytic, cellulolytic and other enzymes (Leinmuller et al., 1991; O’Donovan and Brooker, 2001; Norton and Ahn, 1997).
However, Table 4 indicated that the control group recorded significant (p<0.05) decrease value of ruminal total volatile fatty acids compared with T1 and T2. There are insignificantly (p>0.05) increased in T2 compared with T1. These results are in good agreement with the findings of Yanez Ruiz et al. (2004) who found that increased values of TVFA’s in goats and wethers with PEG addition. Acetate and TVFA’s concentrations increased with PEG supply, reflecting a stimulated microbial activity, as some authors found when treating feedstuffs containing tannins with PEG (Silanikove et al., 1996, 1997). The same effect on TVFA’s and branched-chain VFA (BCVFA) concentrations was observed by McSweeney et al. (2001) using shrub legumes treated with PEG. However, Hassan (2006) noticed increase in TVFA’s of the ruminal fluid in Barki sheep fed on Acacia silage treated with or without PEG. This may suggest that tannins contents had reduced digested nutrients and low fermentable value in control diets than that treated with PEG or Tanozym.
There are insignificant (p>0.05) increase in ruminal fluid NH3 N in T1 and T2 compared with control. These results are in good agreement with the findings of Xu et al. (2007) who found that the rumen ammonia-N contents tended to insignificantly (p>0.05) decrease with increasing level of WGTG (high tannins). Hassan (2006) noticed increase in NH3 N of the ruminal fluid in Barki sheep fed on Acacia silage treated with or without PEG. This may be tannins inhibit the degradation of dietary protein in the rumen and decrease ruminal ammonia concentrations which may suggest an inhibition of proteolytic enzymes in the rumen (Norton and Ahn, 1997). Reduction in protein degradation is associated with a lower production of ammonia nitrogen (Barry and Manley, 1984; Waghorn et al., 1994; Waghorn, 1996).
Blood serum parameters: Table 5 showed blood serum parameters as affected by addition of Tanozym and PEG for Baladi goats diets. Serum total protein in the T2 was significantly (p<0.05) increased compared with control but there were no significant (p>0.05) differences between (T1 and T2) and (control and T1). This may be attributed to the improvements occurred in metabolic process as a response to the PEG and Tanozym additive and indicate that these goats cover their protein needs. These results were agreed by Abdel-Halim (2003) and Kholif et al. (2005), who found that there was a decrease in blood total proteins in goats fed diets containing tannins than control. In addition, serum albumin was a significant (p<0.05) increase in serum albumin by supplementing PEG and Tanozym in goat diets compared with control.
| Table 5: | Effect of experimental diets feeding on some blood serum parameters of lactating goats |
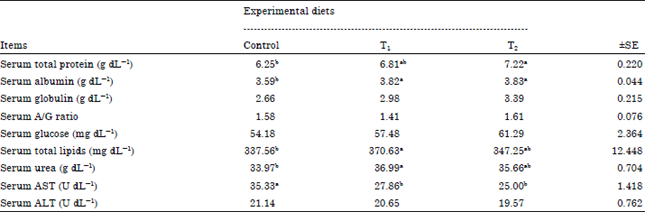 | |
| a,b,cMeans with different superscripts are significant (p<0.05). SE: Standard error | |
This increase may be due to increase in Digestible Crude Protein (DCP). These results are in agreement with those of Kholif et al. (2005) who observed decreasing in blood content of albumin in the ration of tannins content by-products compared with ensiled ration. While, there are no significant (p>0.05) different among groups for blood serum globulin and serum albumin: Globulin (A/G) ratios, these results are agreement with those of Khattab et al. (2008). Serum glucose concentration was highest (p>0.05) in goat fed on T1 and T2 diets than the goats was fed on control diet. These results may be due to the parallel decrease in TVFA’s of rumen fluid (Table 2), these results are in line with those obtained by Khattab et al. (2008). But serum total lipids showed increase by goats fed diets supplemented with Tanozym and PEG compared with control. Serum urea showed increase by goats fed diets supplemented with PEG and Tanozym compared with control. These results are in agreements with those obtained by Silanikove et al. (1996), Abdel-Halim (2003) and Kholif et al. (2005), who observed reduced serum urea concentration in goats fed diets containing tannins which they attributed to low nitrogen availability. Serum aspartate aminotransferase (AST) and alanin aminotransferase (ALT) showed increase by goats fed in control diet compared with T1 and T2. Increasing the control groups may be increase the tannins contents in these diets which effects liver functions (Zhu et al., 1992; Plumlee et al., 1998). But all values of blood parameters are within the normal range.
Milk yield and composition: Data of milk composition and constituent yields of the experimental goats are summarized in Table 6. Milk yield was significantly (p<0.05) increased with Tanozym supplementation to diets of lactating goats. On the other hand, goats fed diets supplemented with Tanozym (T2) showed insignificant (p<0.05) increase milk yield compared to those fed diets supplemented with PEG (T1). The increase of milk yield rate was 5.3 and 9.2% for T1 and T2 compared with control. These results are in good agreement with the findings of Clark et al. (1977) who claimed a positive relationship between blood serum glucose and milk yield. This reduction may by due to one or more of the following reasons; higher TVFA’s content in rumen of animal given control ration (Table 3) or/and lower of nutrients digestibilities, TDN and DCP of control group (Table 2). These results are in agreement with Wang et al. (1996), Gilboa et al. (2000) and Decandia et al. (2000) who found that the high concentrations of tannins caused a decrease in milk yield and digestibility coefficients.
| Table 6: | Effect of experimental diets feeding on milk production and composition |
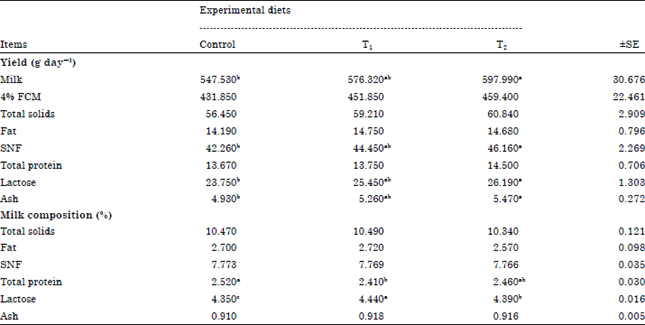 | |
| a,b,cMeans with different superscripts are significant (p<0.05), SE: Standard error | |
Also, 4% FCM yield was increased (p>0.05) in T1 and T2 by about 4.63 and 6.38% than the control. This result agreed with the findings of Khattab et al. (2008).
There were no significant (p>0.05) differences among groups in the percentage of total solids, Solids Not Fat (SNF), fat and ash content among group. This result may be due to the higher milk yield for goats fed diets supplemented with PEG and Tanozym than those fed control diet. These results are agreed by Khattab et al. (2008). While, there were significant (p<0.05) differences among control and T1 but there were insignificant (p>0.05) differences among (control and T2) and (T1 and T2) in the percentage of milk protein but there were significant (p>0.05) differences among groups in the percentage of milk lactose. Milk lactose percentage tended to be lower for goats fed control diets.
CONCLUSION
From the results of this study, it is found that adding Tanozym to reducing tannins effect as anti-nutritional factors of lactating Baladi goats diets led to a significant increase in nutrients digestibility, nutritive values and production performance of lactating goats without any adverse affect on animal health. While, adding of PEG had led to a slight improvement in the performance of lactating goats compared with untreated diets (control). According to those results, adding Tanozym was superior over PEG for improving feed digestion and milk production by Baladi goats.
REFERENCES
- Angaji, L., M. Souri and M.M. Moeini, 2011. Deactivation of tannins in raisin stalk by polyethylene glycol-600: Effect on degradation and gas production in vitro. Afr. J. Biotechnol., 10: 4478-4483.
Direct Link - Barry, T.N., T.R. Manley and S.J. Duncan, 1986. The role of condensed tannins in the nutritional value of Lotus pedunculatus for sheep. 4. Sites of carbohydrate and protein digestion as influenced by dietary reactive tannin concentration. Br. J. Nutr., 55: 123-137.
CrossRefPubMedDirect Link - Clark, J.H., H.R. Spires, R.G. Derrig and M.R. Bennink, 1977. Milk production, nitrogen utilization and glucose synthesis in lactating cows infused postruminal with sodium caseinate and glucose. J. Nutr., 107: 631-644.
Direct Link - Curiel, J.A., H. Rodriguez, I. Acebron, J.M. Mancheno, B. de Las Rivas and R. Munoz, 2009. Production and physicochemical properties of recombinant Lactobacillus plantarum tannase. J. Agric. Food Chem., 57: 6224-6230.
CrossRefPubMedDirect Link - Decandia, M., M. Sitzia, A. Cabiddu, D. Kababya and G. Molle, 2000. The use of polyethylene glycol to reduce the anti-nutritional effects of tannins in goats fed woody species. Small Rumin. Res., 38: 157-164.
CrossRefDirect Link - Dong, Y., H.D. Bae, T.A. McAllister, G.W. Mathison and K.J. Cheng, 1999. Effects of exogenous fibrolytic enzymes, α-bromoethanesulfonate and monensin on fermentation in a rumen simulation (RUSITEC) system. Can. J. Anim. Sci., 79: 491-498.
CrossRefDirect Link - Doumas, B.T., W.A. Watson and H.G. Biggs, 1971. Albumin standards and the measurement of serum albumin with bromcresol green. Clin. Chim. Acta, 31: 87-96.
CrossRefPubMedDirect Link - Hagerman, A.E. and L.G. Butler, 1978. Protein precipitation method for the quantitative determination of tannins. J. Agric. Food Chem., 26: 809-812.
CrossRefDirect Link - Fawcett, J.K. and J.E. Scott, 1960. A rapid and precise method for the determination of urea. J. Clin. Pathol., 13: 156-159.
CrossRefPubMedDirect Link - Fondevila, M. and B. Perez-Espes, 2008. A new in vitro system to study the effect of liquid phase turnover and pH on microbial fermentation of concentrate diets for ruminants. Anim. Feed Sci. Technol., 144: 196-211.
CrossRefDirect Link - Forbes, R.M. and W.P. Garrigus, 1948. Application of a lignin ratio technique to the determination of the nutrient intake of grazing animals. J. Anim. Sci., 7: 373-382.
Direct Link - Frutos, P., M. Raso, G. Hervas, A.R. Mantecon, V. Perez Perez and F.J. Giraldez, 2004. Is there any detrimental effect when a chestnut hydrolysable tannin extract is included in the diet of finishing lambs? Anim. Res., 56: 127-136.
Direct Link - Gallup, W.D., C.S. Hobbs and H.M. Briggs, 1945. The use of silica as a reference substance in digestion trials with ruminants. J. Anim. Sci., 4: 68-71.
Direct Link - Gilboa, N., A. Perevolotsky, S. Landau, Z. Nitsan and N. Silanikove, 2000. Increasing productivity in goats grazing Mediterranean woodland and scrubland by supplementation of polyethylene glycol. Small Rumin. Res., 38: 183-190.
CrossRefDirect Link - Giraldo, L.A., M.J. Ranilla, M.L. Tejido and M.D. Carro, 2004. Effect of enzyme application method on in vitro rumen fermentation of tropical forages. J. Anim. Feed Sci. Suppl., 13: 63-66.
Direct Link - Gornall, A.G., C.J. Bardawill and M.M. David, 1949. Determination of serum proteins by means of the biuret reaction. J. Biol. Chem., 177: 751-766.
CrossRefPubMedDirect Link - Hotz, C. and R.S. Gibson, 2007. Traditional Food-processing and preparation practices to enhance the bioavailability of micronutrients in plant-based diets. J. Nutr., 137: 1097-1100.
PubMedDirect Link - Jones, D.E., 1965. Banana tannin and its reaction with polyethylene glycols. Nature, 206: 299-300.
CrossRefPubMedDirect Link - Jones, W.T. and J.L. Mangan, 1977. Complexes of the condensed tannins of sainfoin (Onobrychis viciifolia scop.) with fraction 1 leaf protein and with submaxillary mucoprotein and their reversal by polyethylene glycol and pH. J. Sci. Food Agric., 28: 126-136.
CrossRefDirect Link - Kung, Jr. L., R.J. Treacher, G.A. Nauman, A.M. Smagala, K.M. Endres and M.A. Cohen, 2000. The effect of treating forages with fibrolytic enzymes on its nutritive value and lactation performance of dairy cows. J. Dairy Sci., 83: 115-122.
CrossRefDirect Link - Lekha, P.K. and B.K. Lonsane, 1997. Production and application of tannin acyl hydrolase: State of the art. Adv. Applied Microbiol., 44: 215-260.
PubMedDirect Link - Lu, M.J. and C. Chen, 2007. Enzymatic tannase treatment of green tea increases in vitro inhibitory activity against N-nitrosation of dimethylamine. Process Biochem., 42: 1285-1290.
CrossRefDirect Link - Makkar, H.P.S., M. Blummel and K. Becker, 1995. Formation of complexes between polyvinyl pyrrolidones or polyethylene glycols and tannins and their implication in gas production and true digestibility in in vitro techniques. Br. J. Nutr., 73: 897-913.
CrossRefPubMedDirect Link - Makkar, H.P.S., 2003. Effects and fate of tannins in ruminant animals, adaptation to tannins, and strategies to overcome detrimental effects of feeding tannin-rich feeds. Small Ruminant Res., 49: 241-256.
CrossRefDirect Link - McSweeney, C.S., B. Palmer, D.M. McNeill and D.O. Krause, 2001. Microbial interactions with tannins: Nutritional consequences for ruminants. Anim. Feed Sci. Technol., 91: 83-93.
CrossRefDirect Link - Mondal, K.C., D. Banerjee, M. Jana and B.R. Pati, 2001. Colorimetric assay method for determination of the tannin acyl hydrolase (EC 3.1.1.20) activity. Anal. Biochem., 295: 168-171.
CrossRefPubMedDirect Link - Norton, B.W. and J.H. Ahn, 1997. A comparison of fresh and dried Calliandra calothyrsus supplements for sheep given a basal diet of barley straw. J. Agric. Sci., 129: 485-494.
CrossRefDirect Link - O'Donovan, L. and J.D. Brooker, 2001. Effect of hydrolysable and condensed tannins on growth, morphology and metabolism of Streptococcus gallolyticus (S. caprinus) and Streptococcus bovis. Microbiology, 147: 1025-1033.
Direct Link - Reitman, S. and S. Frankel, 1957. A colorimetric method for the determination of serum glutamic oxalacetic and glutamic pyruvic transaminases. Am. J. Clin. Pathol., 28: 56-63.
CrossRefPubMedDirect Link - Silanikove, N., Z. Nitsan and A. Perevolotsky, 1994. Effect of a daily supplementation of poly(ethylene glycol) on intake and digestion of tannin-containing leaves (Ceratonia siliqua) by sheep. J. Agric. Food Chem., 42: 2844-2847.
CrossRefDirect Link - Silanikove, N., N. Gilboa, I. Nir, A. Perevolotsky and Z. Nitsan, 1996. Effect of a daily supplementation of polyethylene glycol on intake and digestion of tannin-containing leaves (Quercus calliprinos, Pistacia lentiscus and Ceratonia siliqua) by goats. J. Agric. Food Chem., 44: 199-205.
CrossRefDirect Link - Silanikove, N., N. Gilboa and Z. Ntsan, 1997. Interactions among tannins, supplementation and polyethylene glycol in goats given oak leaves: Effects on digestion and food intake. Anim. Sci., 64: 479-483.
CrossRefDirect Link - Theodorou, M.K., B.A. Williams, M.S. Dhanoa, A.B. McAllan and J. France, 1994. A simple gas production method using a pressure transducer to determine the fermentation kinetics of ruminant feeds. Anim. Feed Sci. Technol., 48: 185-197.
CrossRefDirect Link - Tolera, A., K. Khazaal and E.R. Orskov, 1997. Nutritive evaluation of some browse species. Anim. Feed Sci. Technol., 67: 181-195.
CrossRefDirect Link - Trinder, P., 1969. Determination of blood glucose using an oxidase-peroxidase system with a non-carcinogenic chromogen. J. Clin. Pathol., 22: 158-161.
CrossRefPubMedDirect Link - van Soest, P.J., 1994. Nutritional Ecology of the Ruminant. 2nd Edn., Cornell University Press, Ithaca, New York, Pages: 476.
Direct Link - Waghorn, G.C., I.D. Shelton and W.C. McNabb, 1994. Effects of condensed tannins in Lotus pedunculatus on its nutritive value for sheep. 1. Non-nitrogenous aspects. J. Agric. Sci., 123: 99-107.
CrossRefDirect Link - Warner, A.C., 1964. Production of volatile fatty acids in the rumen: Methods of measurement. Nutr. Abstr. Rev., 34: 339-352.
PubMed - Xu, C., Y. Cai, N. Moriya and M. Ogawa, 2007. Nutritive value for ruminants of green tea grounds as a replacement of brewers' grains in totally mixed ration silage. Anim. Feed Sci. Technol., 138: 228-238.
CrossRefDirect Link - Yanez Ruiz, D.R., A. Moumen, A.I. Martin Garcia and E. Molina Alcaide, 2004. Ruminal fermentation and degradation patterns, protozoa population and urinary purine derivatives excretion in goats and wethers fed diets based on two-stage olive cake: Effect of PEG supply. J. Anim. Sci., 82: 2023-2032.
Direct Link - Zhu, J., L.J. Filippich and M.T. Alsalam, 1992. Tannic acid intoxication in sheep and mice. Res. Vet. Sci., 53: 280-292.
PubMedDirect Link








