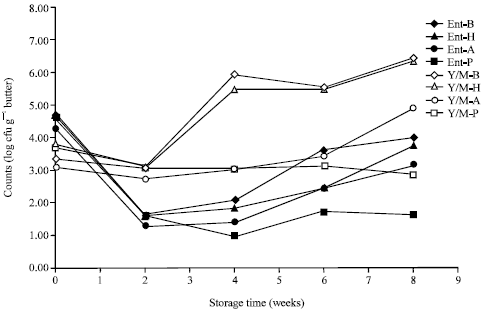
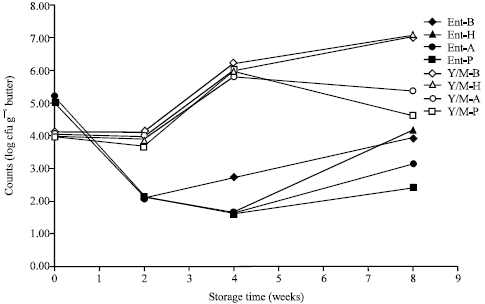
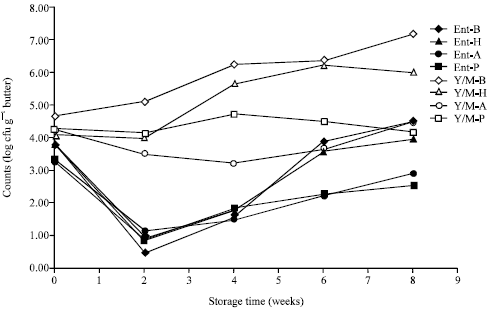
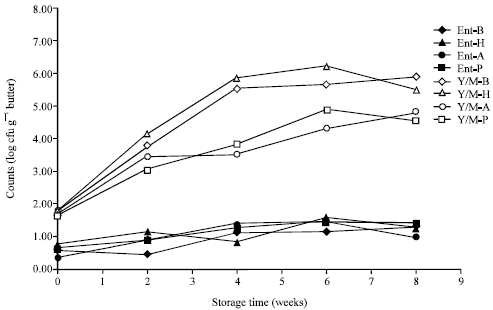
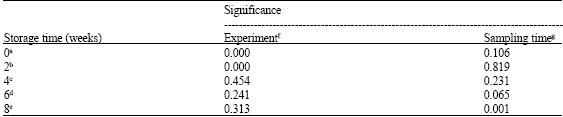
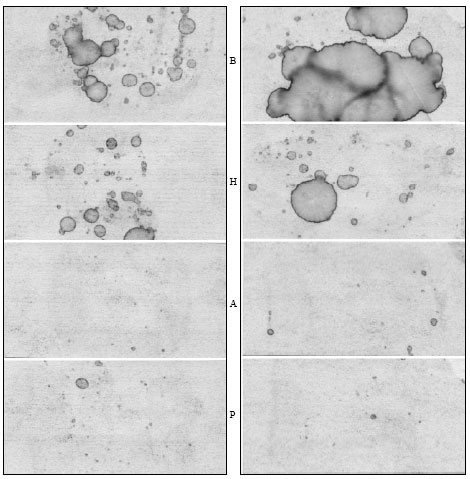
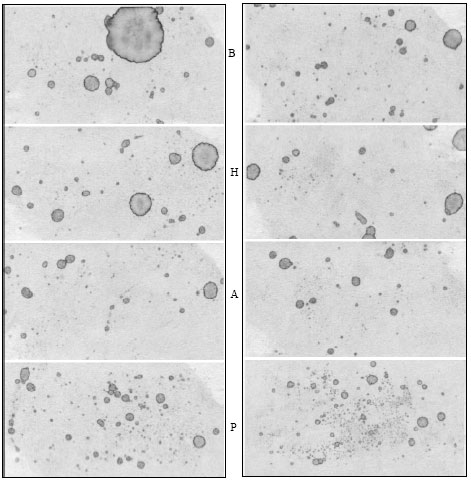
Research Article
Suitability of a Moisture Distribution Assay to Assess the Microbial Stability of Butter Produced at a Small Scale
Laboratory for Food Science, Leuven Food Science and Nutrition Research Centre (LfoRCe), Department of Biosciences and Technology, K.H. Kempen University College, Kleinhoefstraat 4, B-2440 Geel, Belgium
Sophie Schittecat
Laboratory for Food Science, Leuven Food Science and Nutrition Research Centre (LfoRCe), Department of Biosciences and Technology, K.H. Kempen University College, Kleinhoefstraat 4, B-2440 Geel, Belgium
Sofie Vandewijngaarden
Laboratory for Food Science, Leuven Food Science and Nutrition Research Centre (LfoRCe), Department of Biosciences and Technology, K.H. Kempen University College, Kleinhoefstraat 4, B-2440 Geel, Belgium
Johan Claes
Laboratory for Food Science, Leuven Food Science and Nutrition Research Centre (LfoRCe), Department of Biosciences and Technology, K.H. Kempen University College, Kleinhoefstraat 4, B-2440 Geel, Belgium