Research Article
In vitro Seed Germination and Micropropagation of Edible Bamboo Dendrocalamus giganteus Munro using Seeds
Center of Advanced Study in Life Sciences, Takyelpat, Imphal-795001, Manipur, India
L. Bengyella
Department of Biotechnology, University of Burdwan, Golapbag-7131104, West Bengal, India
G.J. Sharma
Center of Advanced Study in Life Sciences, Takyelpat, Imphal-795001, Manipur, India









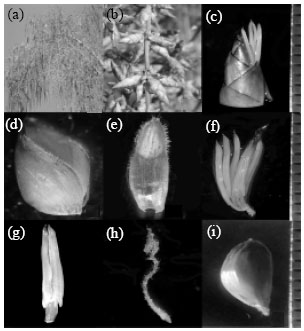
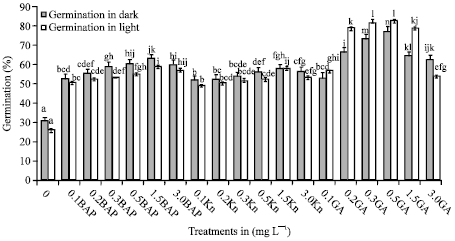
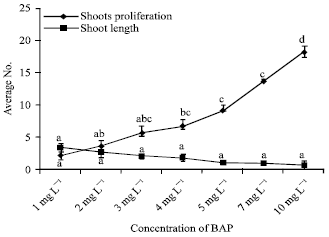
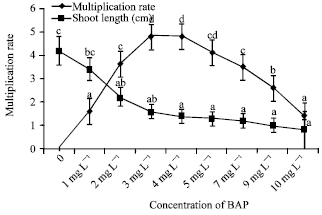

Tahir SM Reply
This is a well researched highly educative paper. Keep it up.
I've learnt a lot.
Thank you
Sayanika DW
Thanks Tahir. It was painful optimizing this work and i am happy you learned something.
Cheers