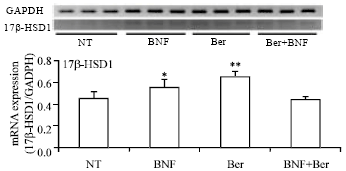
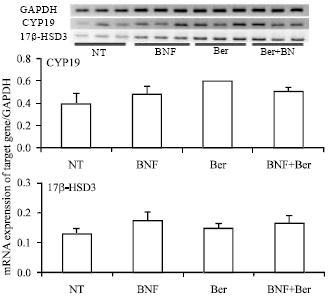
Research Article
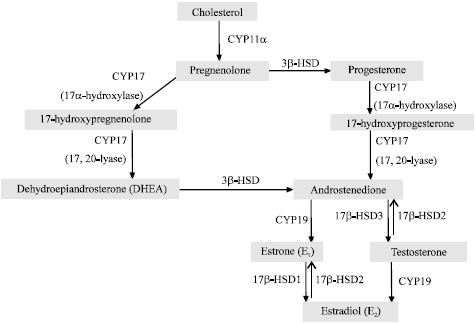
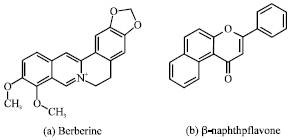
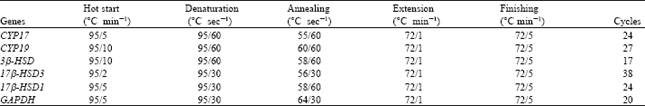
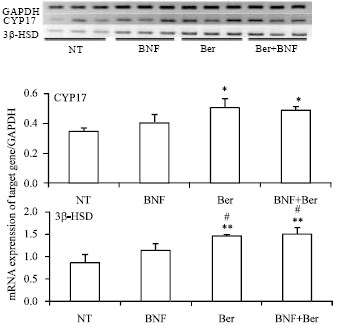
Berberine Disturbs the Expression of Sex-hormone Regulated Genes in β-naphthoflavone-induced Mice
Research Group for Pharmaceutical Activities of Natural Products using Pharmaceutical Biotechnology, Faculty of Pharmaceutical Sciences, National Research University-Khon Kaen University, Khon Kaen, 40002, Thailand
Waranya Chatuphonprasert
Research Group for Pharmaceutical Activities of Natural Products using Pharmaceutical Biotechnology, Faculty of Pharmaceutical Sciences, National Research University-Khon Kaen University, Khon Kaen, 40002, Thailand
Kanokwan Jarukamjorn
Research Group for Pharmaceutical Activities of Natural Products using Pharmaceutical Biotechnology, Faculty of Pharmaceutical Sciences, National Research University-Khon Kaen University, Khon Kaen, 40002, Thailand