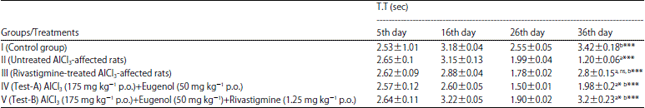
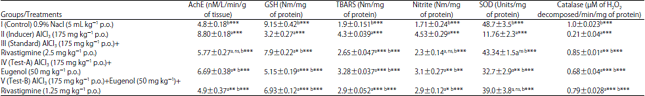
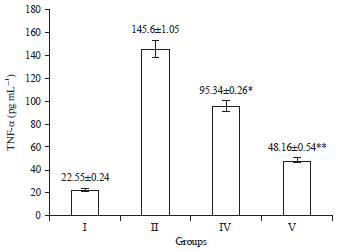
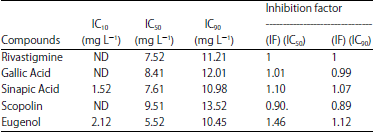Research Article
Modulation of Pro-inflammatory Mediators by Eugenol in AlCl3 Induced Dementia in Rats
Department of Neurology, Jinan Central Hospital Affiliated to Shandong University, No. 105, Jiefang Road, Lixia, Jinan, 250013, Shandong, People Republic of China
Dong Wang
Department of Neurology, Jinan Central Hospital Affiliated to Shandong University, No. 105, Jiefang Road, Lixia, Jinan, 250013, Shandong, People Republic of China
Souravh Bais
Department of Pharmacology, Rayat Institute of Pharmacy, Railmajra, Distt. S.B.S. Nagar, 144533, Punjab, India
LiveDNA: 91.5733
Hongxin Wang
Department of Neurology, Jinan Central Hospital Affiliated to Shandong University, No. 105, Jiefang Road, Lixia, Jinan, 250013, Shandong, People Republic of China
LiveDNA: 86.25486