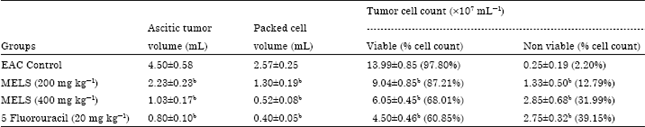
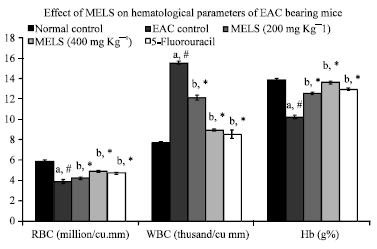
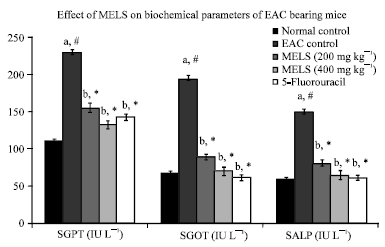
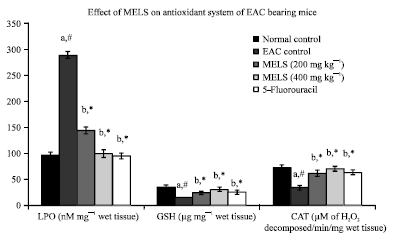
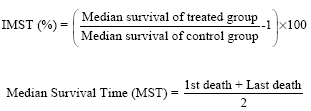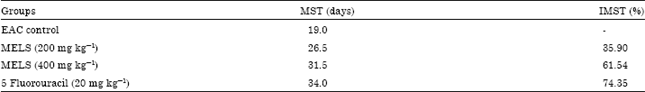Research Article
Evaluation of Anticancer Activity of Lagenaria siceraria Aerial Parts
Guru Nanak Institute of Pharmaceutical Science and Technology, 157/F, Nilgunj Road, Panihati, Kolkata-700114, India
S. Kundu Sen
Department of Pharmaceutical Technology, Jadavpur University, Kolkata-700 032, India
A. Bala
Department of Pharmaceutical Technology, Jadavpur University, Kolkata-700 032, India
U.K. Mazumder
Department of Pharmaceutical Technology, Jadavpur University, Kolkata-700 032, India
P.K. Haldar
Department of Pharmaceutical Technology, Jadavpur University, Kolkata-700 032, India