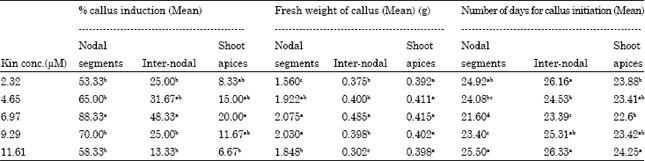
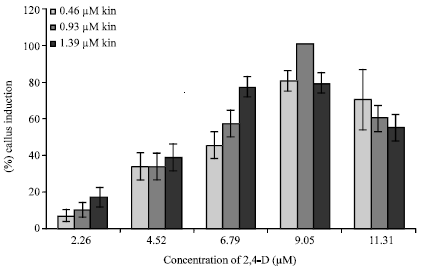
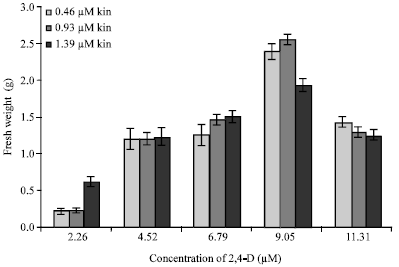
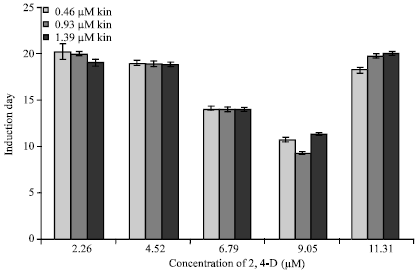
Research Article
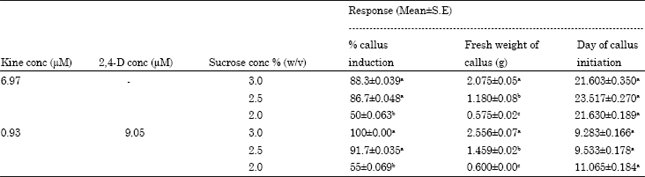
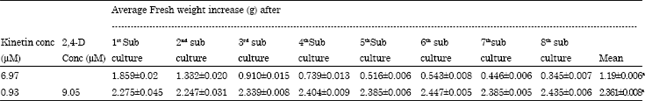
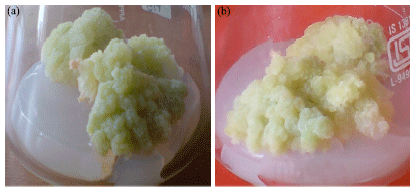
The Influence of Plant Growth Regulators, Explant Nature and Sucrose Concentration on in vitro Callus Growth of Thevetia peruviana Schum
Department of Biotechnology, Chaudhary Devi Lal University, Sirsa, Haryana, India
Kiran Grover
Department of Biotechnology, Chaudhary Devi Lal University, Sirsa, Haryana, India
Anita Rani Gill
Department of Biotechnology, Chaudhary Devi Lal University, Sirsa, Haryana, India