Research Article
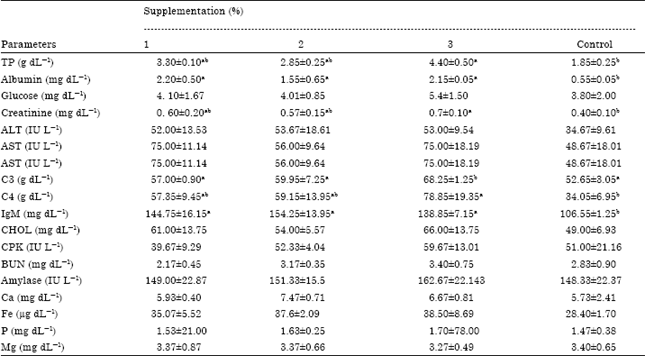
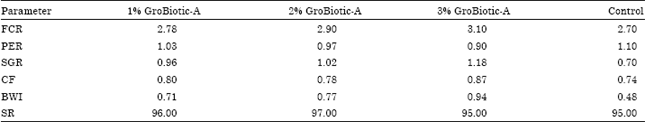
Effect of Prebiotic Supplementation on Growth Performance and Serum Biochemical Parameters of Kutum (Rutilus frisii kutum) Fries
Department of Fishery, Qaemshahr Branch, Islamic Azad University, Qaemshahr, Iran
Masoud Hedayatifard
Department of Fishery, Qaemshahr Branch, Islamic Azad University, Qaemshahr, Iran
Shahram Fahimi
Department of Fishery, Qaemshahr Branch, Islamic Azad University, Qaemshahr, Iran
Mojtaba Shikholeslami
Department of Fishery, Qaemshahr Branch, Islamic Azad University, Qaemshahr, Iran
Mehdad Irani
Department of Fishery, Qaemshahr Branch, Islamic Azad University, Qaemshahr, Iran
Cyrus Amirinia
Department of Fishery, Qaemshahr Branch, Islamic Azad University, Qaemshahr, Iran
Seyed Ehsan Mousavi
Department of Fishery, Qaemshahr Branch, Islamic Azad University, Qaemshahr, Iran