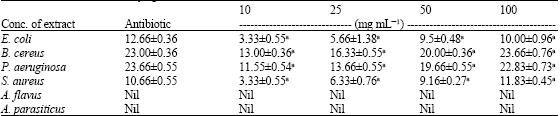
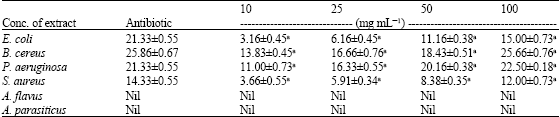
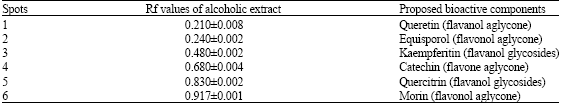
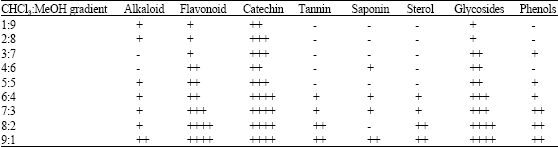
Research Article
Bioassay-Guided Isolation and Identification of Antibacterial and Antifungal Component from Methanolic Extract of Green Tea Leaves (Camellia sinensis)
Ashok and Rita Patel Institute of Integrated Study and Research in Biotechnology and Allied Sciences, New Vallabh Vidya Nagar, Gujarat-300018, India
Sunanda Chakraborti
Ashok and Rita Patel Institute of Integrated Study and Research in Biotechnology and Allied Sciences, New Vallabh Vidya Nagar, Gujarat-300018, India