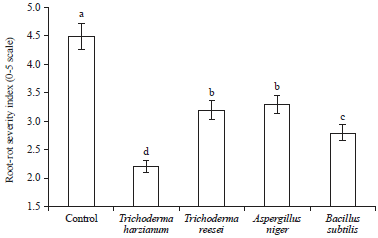
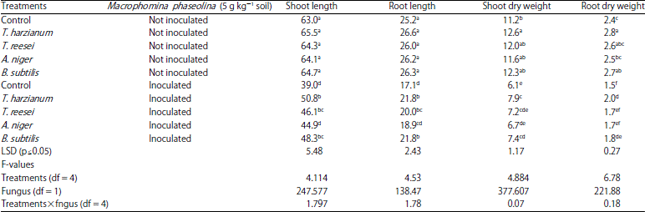
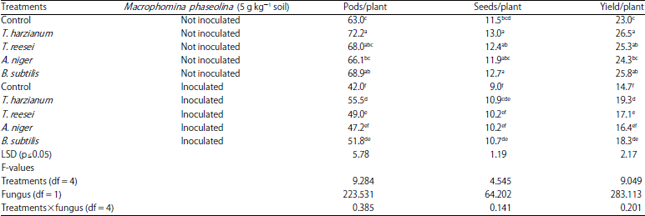
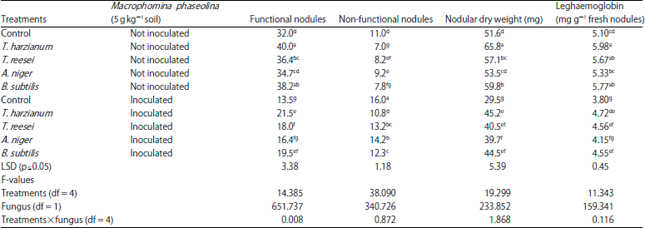
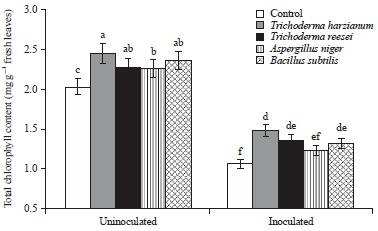
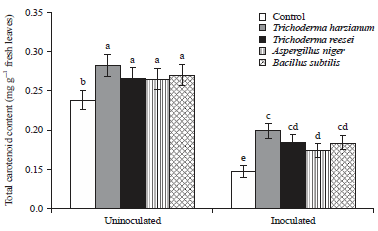
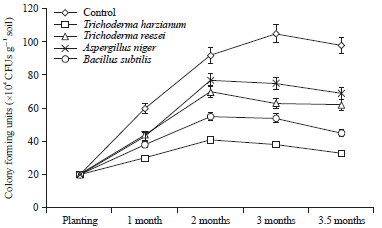
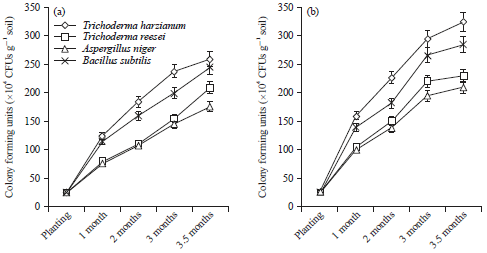
Research Article
Biological Control of Root-rot on Mungbean Plants Incited By Macrophomina phaseolina Through Microbial Antagonists
Department of Plant Protection, Faculty of Agricultural Sciences, Aligarh Muslim University, 202002 Aligarh, India
Mujeebur Rahman Khan
Department of Plant Protection, Faculty of Agricultural Sciences, Aligarh Muslim University, 202002 Aligarh, India