Research Article
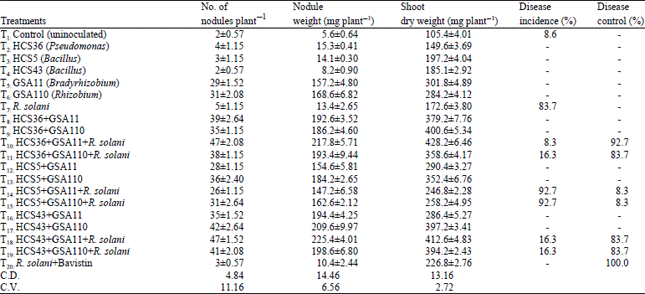
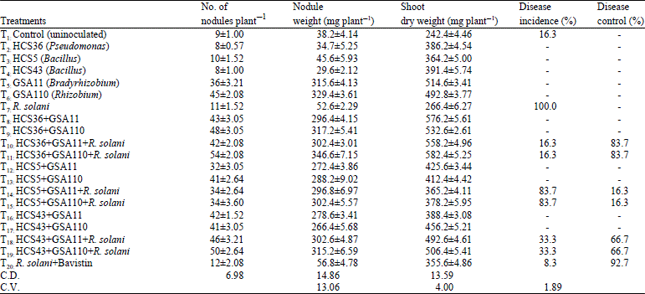
Suppression of Rhizoctonia solani Root Rot Disease of Clusterbean (Cyamopsis tetragonoloba) and Plant Growth Promotion by Rhizosphere Bacteria
Department of Microbiology, College of Basic Sciences and Humanities, CCS Haryana Agricultural University, Hisar, 125 004, India
Satyavir Singh Sindhu
Department of Microbiology, College of Basic Sciences and Humanities, CCS Haryana Agricultural University, Hisar, 125 004, India