Research Article
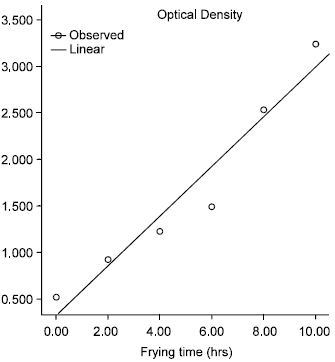
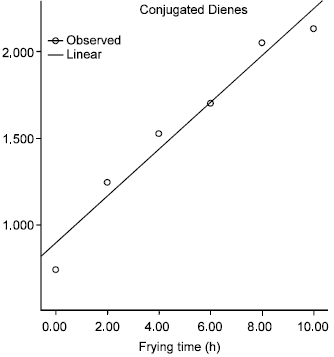
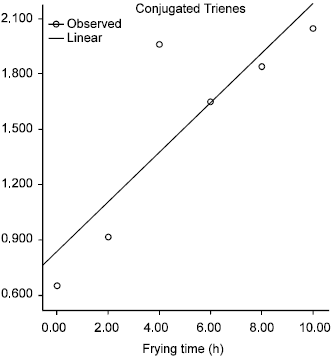
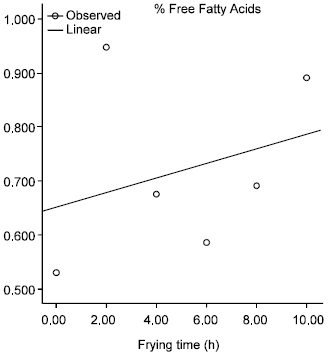
Effect of Commercial Kebab Frying on Physico-Chemical Parameters of the Tallow
Department of Biotechnology, University of Malakand, Chakdara, NWFP, Pakistan
Ikram Ullah
Department of Biotechnology, University of Malakand, Chakdara, NWFP, Pakistan
Saeed Ahmad
Department of Biotechnology, University of Malakand, Chakdara, NWFP, Pakistan
Hayat Khan
Department of Biotechnology, University of Malakand, Chakdara, NWFP, Pakistan
Haji Akbar
Department of Biotechnology, University of Malakand, Chakdara, NWFP, Pakistan