Research Article
Lipid Profile of Rats Fed Cholesterol, Barely and Wheat
Department of Nutrition and Food Technology, Faculty of Agriculture, Mu`tah University, Mu`tah, Al-Karak, P.O. Box 7, Jordan
Cholesterol homeostasis is maintained and carefully controlled by multiple feedback mechanisms. It regulated by the interaction of three factors: absorption, synthesis and excretion. Cholesterol may be obtained for cellular metabolism either via uptake endogenous and exogenous (dietary) cholesterol or through biosynthesis (Tabas, 2002; Okazaki et al., 2006; Torres-Gonzalez et al., 2007). Although cholesterol uptake and biosynthesis are interdependent, cellular cholesterol requirements can be met equally well through either supply pathway. Human body synthesizes approximately 700-900 mg of cholesterol per day, while 300-500 mg are absorbed daily from dietary sources. Normal daily cholesterol turnover is accounted for by excretion in the gastrointestinal tract (600 mg/day), conversion to bile acids (400 mg/day), sloughing skin (85 rag/day), biosynthesis of steroid hormones (50 mg/day) and by incorporation into membranes of actively dividing cells (Russell, 1992). Dietary cholesterol affects concentrations of Total Plasma Cholesterol (TC) and Low Density Lipoprotein cholesterol (LDL-c) in humans and animals. Feeding of cholesterol-enriched diet resulted in hypercholesterolemia characterized by increased LDL-c and Very Low Density Lipoprotein cholesterol (VLDL-c) and a concomitant decrease of High Density Lipoprotein Cholesterol (HDL-c) in, susceptible white rabbits to the development of hypercholesterolemia, (Overturf et al., 1990). This diet commonly has similar effects in other animal species as well. It showed in rats (Mahley and Holcombe, 1977), dogs (Mahley et al., 1974), monkeys (Lusk et al., 19982; Hunt et al., 1992) and to a lesser extent, humans (Bersot et al., 1986). An elevated TC concentration and especially, LDL-c, increases the risk of Coronary Heart Disease (CHD) and atherosclerosis (Rastogi et al., 2004; Sanchez-Muniz et al., 2009). Vegetarians are known to have low plasma total and LDL-c concentrations, which are related to low intake of dietary cholesterol and fat and to high dietary fiber content (Nestel et al., 1981; Vuoristo and Miettinen, 1994). Further, studies have reported that lacto-ovo-vegetarians have low plasma cholesterol concentrations and desirable lipoprotein profiles (Masarei et al., 1984), which suggests that consuming relatively high amounts of dietary cholesterol with high amounts of vegetable protein and adequate amounts of dietary fiber will not result in elevated plasma cholesterol concentrations. (Jenkins et al., 1997; Fernandez et al., 1999). The best "support" for the idea that a high cholesterol diet leads to coronary artery disease in human beings comes from the statements that coronary artery disease is rare in Chinese because such individuals do not ingest cholesterol in great amounts (Vuoristo and Miettinen, 1994). The atheromatous plaque development can be slowed or prevented with lipid lowering came initially from animal studies on cholesterol-fed primates and histological measurement of plaque size following dietary restriction. (Andersen et al., 1987; Wissler and Vesselinovitch, 1990).
Cholesterol absorption from the intestinal milieu occurs through a series of steps, involving emulsification, hydrolysis, micellar transport, mucosal absorption, resynthesis of parent molecules in enterocytes and assembly with apolipoproteins to form chylomicrons, which are secreted from intestinal cells (Lu et al., 2001). Some or all of these steps can be interfered with by dietary components and drugs (Ros, 2000; Thomsen et al., 2004). Both drugs and diet have been shown to be effective in reducing blood cholesterol level and CHD risk and mortality (Lin et al., 2004; Jenkins et al., 2006). Statin drugs provide effective cholesterol lowering therapy and are widely prescribed, but they can cause severe adverse effects (McKinney et al., 2006). Lowering plasma LDL-c through no drug strategies, such as consuming specific food components such as plant sterols and soy protein, would therefore be most desirable (Weststrate and Meijer 1998; Teixeira et al., 2000; Rasmussen et al., 2006). To increase the relevance of dietary advice for the primary prevention of cardiovascular disease, the National Cholesterol Education Program (NCEP, 2001) has recommended the use of functional foods or foods high in components that reduce cholesterol as additional options to enhance the effectiveness of cholesterol-lowering diets. These functional ingredients include fibers, soy protein, plant sterols and nuts. The US Food and Drug Administration now allow foods with these components to carry a health claim indicating that they reduce the risk of cardiovascular disease (FDA, 2003). The consumption of dietary fiber, especially water-soluble fiber, has been shown to be inversely associated with CHD. The heart-healthy diet proposed by the Canadian Heart and Stroke Foundation recommends consumption of 21-38 g/d of fiber and suggests that inclusion of soluble fiber may help lower cholesterol and blood sugar (Ames and Rhymer, 2008). Similar dietary fiber guidelines followed by NCEP (2001). Barley foods could help consumers achieve these dietary goals. The ability of barley β-glucan soluble fiber to lower serum cholesterol is thought to occur through a combination of factors and mechanisms. Suggested mechanisms of cholesterol reduction after increased soluble fiber consumption include delayed intestinal absorption of glucose and lipids and inhibition of absorption and reabsorption of cholesterol and bile acids accompanied by increased excretion of bile acids (Wilson et al., 2004). The reduced absorption may be caused by the high viscosity of β-glucan solutions, which increase the viscosity of the intestinal contents (Jenkins et al., 2002). Other factors may also be important, such as the fermentation of β-glucan in the colon, resulting in production of short-chain fatty acids, which impede cholesterol biosynthesis (Wilson et al., 2004).
Research studies examining foods are important, because they account for biological interactions that might otherwise be lost in the analysis of individual nutrients. Single-nutrient studies are also needed to explore the mechanisms by which foods may be protective. The objective of current study was to examine associations between whole barely and wheat grains and exogenous cholesterol and coronary heart disease risk factors.
MATERIALS AND METHODS
Animals: One hundred and five albino Sprague-Dawley derived male rats, weighting 62±6 g (3 week old) were housed individually in stainless steel cages having wires mesh bottoms and fronts in an environmentally controlled room maintained at about 22-25oC and a lightness between 8:00 am to 8:00 pm daily. Rats were randomly divided into 15 groups (7 rats each) and fed with ad libitum access for 28 days, Food consumption of each rat was recorded daily. The animals were weighted once a week. Food scrap was weighted daily. Feces were collected during the last 3 d of the experimental period, dried and stored at -18oC. At the conclusion of experiment, rats were deprived of feed for 12 h, weighted and anesthetized with chloroform. Blood was withdrawn by cardiac puncture and rats were killed by exsanguinations. Blood samples were centrifuged and serums were collected. The liver, heart, kidneys and spleen were excised, blotted and weighed. Serum and liver samples were stored at -18oC until analysis.
| Table 1: | Analysis of barley and wheat |
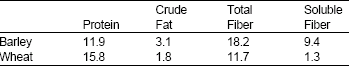 | |
Diets: In brief, the diets were based on casein, cornstarch, corn oil and sucrose and contained added vitamins and minerals. The diets contained additionally finely ground barley and wheat. Both cereals were milled to uniform size (to pass a 2-mm mesh) and added to provide 100 g of total fiber/kg diet. Corrections were made in the quantities of casein, corn oil and starch for the protein, fat and carbohydrates present in barley and wheat so that all diets contained the same concentrations of fat, protein and carbohydrate. At the beginning, barley, wheat and casein were analyzed. The analytical data are shown in Table 1. Control diet (basal diet) contained casein- vitamins free (BDH Chem. Ltd. Co., England), corn starch, corn oil, sucrose (from local market of Al-karak city) and vitamin and mineral mixtures as indicating latter. These ingredients were formulated to make 1 kg of (basal diet). Milled barley (Hordeum vulgare L., RUM) and wheat (Triticum aestirum L., HORANI 27) from local market of Al-karak city were added to the basal diet to make 1 kg of (fiber diet). Cellulose (Merchary Nagel and Co. D-516 D Uren- Germany) was added to the wheat diet to obtain isofiber. Cholesterol (Sigma Chem. Com. St. Louis, MO) was added at four levels (1, 2, 3 and 4 g) to the 1 kg of basal diet and of fiber diets.
| Table 2: | Composition of diets (g/kg) fed to the rats for 28 days* |
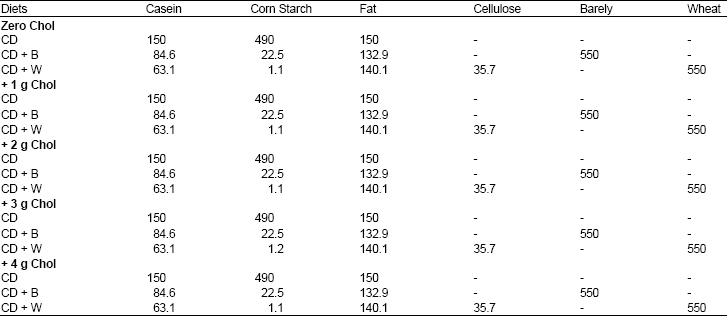 | |
*Chol: cholesterol. CD: control diet. B: Barely. W: Wheat. (150 g) of sucrose, (50) of mineral mixture and (10) of vitamin mixture were added to these diets. Mineral mixture contained (g/kg): FeSO4.7H2O, 3.5; KCL, ZnSO4, 0.2; MnSO4.H2O, 2.0; CuSO4, 4.0; KI, 0.08; MgCO3, 28 and dextrose to make 1 kg mixture. Vitamin Mixture contained (g/kg): vitamin A (retinal acetate), 6.88 ; vitamin D3 (calciferol), 0.125; alphatochopherol, 4.0; thiaminhydrochloride, 1.0; riboflavin, 4.0; pyridoxine hydrochloride, 3.0; niacin, 10.0; folic acid, 0.5; calcium pantothenate, 5.0; vitamin B12, 0.005; choline hydrochloride, 75.0; ascorbic acid, 45.0; biotin, 0.02; menadion, 125.0 and dextrose to make 1 kg mixture | |
All diets were balanced to contain 15% protein, 15% fat (about 25% of total diet energy). 10% total dietary fiber. Grains were included in fiber diets at a 55% level. The diets were manufactured by mixing dry components and oil in a stainless steel commercial dough mixer. The thoroughly mixed components were then moistened with distilled water, pasted, pelted (about 5 g each) and air-dried overnight at 40-45oC. Diets were prepared 2 d before the start of the experiment. The pellets were stored in air-tight drums at 0-4oC. Table 2 shows the 15 diets components.
Analysis: Crude fat in barley and wheat measured by Soxtec System HT (Tecator, Inc., Herndon, VA). Protein content in wheat, barley and casein measured by the Kjeldahl method of AOAC, sec. 7.015 (1980). Fiber content in barley and wheat was measured by the Prosky et al. (1988) method. Serum Total Lipid (TL) concentration was measured according to the method of Sperry and Brand (1995). Samples of diet, liver, heart, kidneys, spleen and feces were mixed with sand and homogenized using a mixer and Total Lipid (TL) was extracted with chloroform/methanol (2/1, v/v) according to the method of Folch et al. (1957). The percent of TL was measured gravimetrically and these lipid extracts were diluted with chloroform to make 5 ml for next analysis. The TC concentration in serum and lipid extracts of endogenous organs and feces was determined using the enzymatic method from Arab Company for medical diagnostic. Triglycerides (TG) concentration in serum and liver was determined using the enzymatic method from Bicon, Germany. Concentrations of Free Cholesterol (FC) and Phospholipids (PL) in the serum and liver were measured according to methods mentioned by Tietz (1987). Cholesterol Ester (CE) concentrations in the serums and liver were calculated from the difference between TC and CE. Concentration of HDL-c in the serum was measured by the precipitation technique using magnesium chloride and phosphotungstic acid from Bicon, Germany. Concentration of serum LDL-c was obtained as the difference between TC and HDL-c using the formula of Friedewald et al. (1972), LDL-c = (TC) - (HDL-c) - (TG/5).
Determination of neutral sterols and bile acids in rat feces: collected rat feces were dried and grounded. About one gram from the ground feces was used to extract both neutral sterols and bile acids and then analyzed quantitatively using Hawlet-Packard gas chromatography according to the method of Reddy et al. (1977). Neutral sterols and bile acids were analyzed as trifluoroacetyl derivatives on a hybrid column packed with 3% QF-1 (Supelco, Inc.). The identification of neutral sterols and bile acids was made by using standard references.
The data were expressed as Mean±Standard Deviation (SD). Statistical differences were determined by Duncan's multiple range tests at p<0.05 by SAS Version (1988).
Table 3 presents the daily food intake, calculated energy intake, weight gain of rats and liver weight. The data revealed that mean of daily food consumption by rat group fed basal diet was higher than that by rat groups fed barely and wheat containing diets.
| Table 3: | Food Intake (FI, g), calculated Energy Intake (EI, kcal), Initial Weight (IW, g), body Weigh Gain (WG, g) and % of weight of liver, heart, kidneys and spleen to final total body weight (FW) of rats fed Cholesterol (Chol), Barely (B) and Wheat (W) |
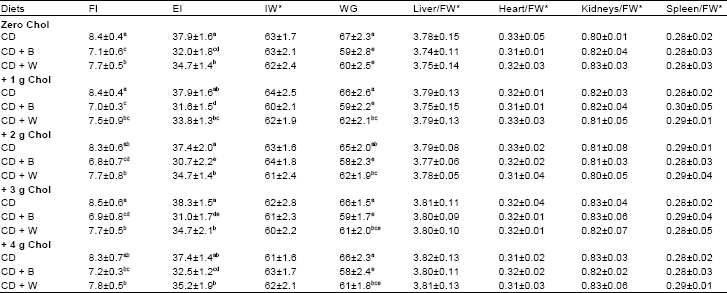 | |
CD: Control Diet: *No significant differences in the data. Values are means±SD. Means in a column without a common letter differ, p>0.05 | |
The lowest daily food intake (about 7 g) was detected in rat groups fed barely containing diets. Consequently, calculated energy intake had the same trend, whereas control rats received the highest energy intake (about 38 kcal) followed by rat groups fed wheat containing diets (about 35 kcal). At the end of feeding course, means of body weight gains of rat groups fed wheat and barely containing diets were lower than that of rat groups fed control diet. Diet type had no significant effect on the percents of weight of liver, heart, kidneys and of spleen to final body weight. However, added cholesterol to the rat diets had no effects on their daily food intake, weight gain and weights of endogenous organs/body weight.
Table 4 shows the effects of dietary cholesterol, barely and wheat on TL and TC of rats' serum, liver, heart, kidneys spleen and feces.
Cholesterol-free diets: The levels of TL and TC in serum (mg/100 ml), liver, heart, kidney and spleen (mg/g dry wt) of rats fed diet containing barely were significantly lower than those found in rats fed diet containing wheat and control diet. Meanwhile, rats fed control diet had the highest values of TL (331, 72.2, 23.2, 26.1 and 12.0) and of TC (81.2, 5.2, 2.6, 3.2 and 2.9) in the serum liver, heart, kidney and spleen, respectively, followed by wheat diet. Barely diet resulted in the highest level of feces TL (218) and TC (6.4).
Cholesterol containing diets: When cholesterol was added to the basal diet resulted in insignificantly increases in TL and significant increase in TC in serum, liver, heart and kidney, spleen and feces. The highest values of serum, liver, heart, kidney and spleen TC (88.7, 6.4, 3.3, 3.8, 3.4 and 6.0, respectively) was observed in rats fed basal diet +4 g cholesterol. Effects of added barely and wheat to this diet had the same observed trend when added to the cholesterol-free basal diet. It caused significantly reductions in lipids of serum and endogenous organs accompanied by significantly increase in fecal lipids. The reduction effects of barely were significantly higher than that of wheat. Cholesterol effects with barely and wheat containing diets had the same trend showed with basal diet. It resulted (at high doses 3 and 4 g cholesterol) in significantly raising effect in serum and liver TC.
Table 5 shows the levels of TG, PL, FC, CE and CE/TC (%) in serum and liver of rats fed dietary cholesterol, barely and wheat.
Cholesterol-free diets: The data showed that the levels of TG, FC and CE in serum and liver of rats fed barely containing diet were significantly lower than those in rats fed wheat containing diet and control diet. The highest levels of serum (mg/100 ml) and liver (mg/g dry wt) TG (132 and 19.4), FC (20.8 and 3.4) and CE (60.4 and 1.8) were showed in rats fed control diet, followed by rats fed wheat diet. There were no significant effect to the diet type on serum and liver PL. Barely diet resulted in significantly reduction in percent of CE/TC in rats' serum and liver compared with wheat and control diets.
Cholesterol containing diets: Added cholesterol to the rat diets resulted in no significant increase in serum and liver TG (only in rats fed control diet with high dose of cholesterol, +4 g, the increase in liver TG was significant compared with rats fed with low dose of cholesterol), whereas the significant effects of dietary cholesterol were showed in serum and liver FC and CE. Dietary cholesterol had no significant effects in serum and liver PL and serum CE/TC, whereas resulted in significant reduction in liver CE/TC in rats fed barely and control diets but not wheat diet.
| Table 4: | Total lipids and total cholesterol in serum (mg/100 ml) and in liver, heart, kidneys, spleen and feces (mg/g dry wt) of rats fed Cholesterol (Chol), Barely (B) and Wheat (W) for 28 days |
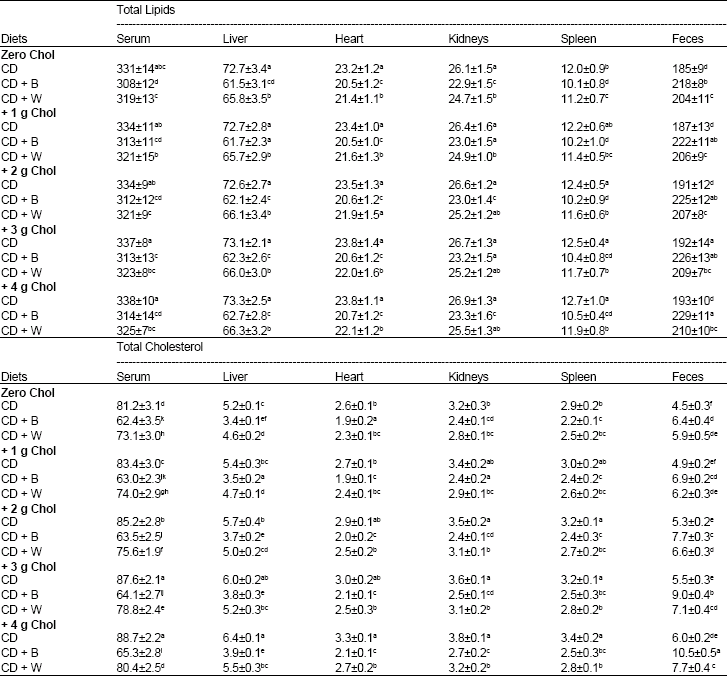 | |
CD: Control Diet. Values are means±SD. Means in a column without a common letter differ, p>0.05 | |
Table 6 shows the levels of LDL-c and HDL-c and LDL-c/ TC and LDL-c/HDL-c in serum of rats fed cholesterol, barely and wheat.
Cholesterol- free diets: The data showed that the effects of control, barely and wheat diets in serum LDL-c and HDL-c were followed the significant decreasing order: barely > wheat > control. However, rats fed control diet had the highest serum (mg/100 ml) LDL-c (39.2) and HDL-c (15.6). Barely containing diet resulted in the lowest atherogenic indices, LDL-c/ TC (44.1%) and LDL-c/ HDL-c (243%) in rat serum compared with wheat and control diets.
Cholesterol containing diets: Dietary cholesterol resulted in significantly increase in serum LDL-c and HDL-c of rats fed control and wheat diets. With barely containing diets, added cholesterol had no significant raising effects in these parameters. The highest serum (mg/100 ml) LDL-c (44.0) and HDL-c (17.7) levels were showed in rats fed control diet +4 g cholesterol. However, dietary cholesterol resulted in no significant increase in atherogenic indices in serum of all rat groups.
| Table 5: | Triglycerides (TG), Phospholipids (PL), Free Cholesterol (FC), Cholesterol Ester (CE) and CE/ Total Cholesterol (TC) in serum (mg/100 ml) and liver (mg/g dry wt) of rats fed Cholesterol (Chol), Barely (B) and wheat (W) for 28 days |
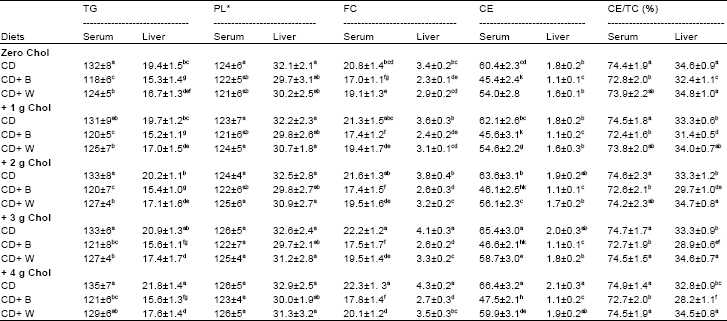 | |
CD: Control Diet. *The effect of diet type in serum PL/TG ratio was followed the order: CD+B > CD+W > CD. Values are means±SD. Means in a column without a common letter differ, p>0.05 | |
Table 7 shows the influences of dietary cholesterol, barely and wheat on rats' fecal neutral sterols.
Cholesterol- free diets: Among rats fed cholesterol-free diets, total fecal neutral sterols (mg/g dry wt) was insignificantly higher (8.39) in rat group fed barely containing diet than in rat group fed wheat containing diets (7.50) and in rat group fed basal diet (7.24).
Cholesterol containing diets: When rats were fed cholesterol containing diets, all rat groups had significantly higher total fecal excretion of neutral sterols, in dose-dependant manner, than did the same diets without cholesterol. Rat groups fed, with cholesterol, barely and wheat containing diets with cholesterol resulted in higher total fecal neutral sterols than rat groups fed basal diet with cholesterol. Barely effect in excretion of exogenous cholesterol was higher than that of wheat. The highest (17.63) total neutral fecal sterols (mg/g dry wt) was observed in rat group fed barely containing diet +4 g cholesterol.
Table 8 shows the influences of dietary cholesterol, barely and wheat on rats' bile acids excretion.
Cholesterol-free diets: Barely containing diet resulted in significantly and insignificantly higher rat fecal bile acids (mg/g dry wt) excretion (12.98) compared with control diet (9.92) and wheat containing diet (11.14), respectively.
Cholesterol containing diets: The addition of cholesterol to the rat diets caused significant increase in bile acid excretion in dose dependant manner. This effect was showed with all tested diets. However, with barely containing diet the effect of added cholesterol in bile acids excretion was significantly higher than that with wheat containing diet and control diet, in other words, the effect of exogenous cholesterol in excretion of rats bile acids was followed the order: with barely diet > with wheat diet > with control diet. The highest and lowest fecal bile acids (mg/g dry wt) levels were showed in rats fed barely containing diet +4 g cholesterol (25.35) and rats fed control diet + 1 g cholesterol (11.04).
The use of a small animal model in a controlled environment is logical to evaluate dietary ingredients. Among the rodents, rats have been shown to have a good response to dietary factors (Tsuda et al., 1983). Randomization of rats into 15 experimental groups resulted in initial body weights that were not significantly different. Rat groups fed barely and wheat containing diets had lower daily food intake than those fed control diet. This lower food intake was reflected by a significantly lower mean body weight gain. A diet which is high in dietary fiber is generally bulky and is less likely to cause overeating and weight gain. Rats seemed to eat less after eating high satiety diets (fiber containing diets) than after eating low satiety diet (fiber free diet). Dietary fiber has a high water holding capacity (Eastwood, 1973) and it is likely that rats fed fibers, in spite of the lower energy in their diet compared to those fed fiber- free diet, did not eat more because the fiber is swelling in their intestine and gives satiety sense. As a result rats fed fiber diets utilized more fats from their food, while the control rats deposited it in their tissues.
| Table 6: | Low density lipoprotein-cholesterol (LDL-c), high density lipoprotein-cholesterol (HDL-c), LDL-c/ total cholesterol (TC) and LDL-c/HDL-c in serum of rats fed cholesterol (Chol), barely (B) and wheat (W) for 28 days |
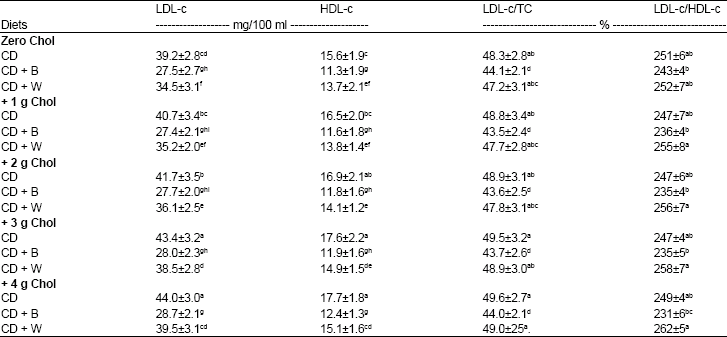 | |
CD: Control Diet. Values are means±SD. Means in a column without a common letter differ, p>0.05 | |
| Table 7: | Neutral sterols in feces (mg/g dry wt) of rats fed Cholesterol (Chol), Barely (B) and wheat (W) for 28 days |
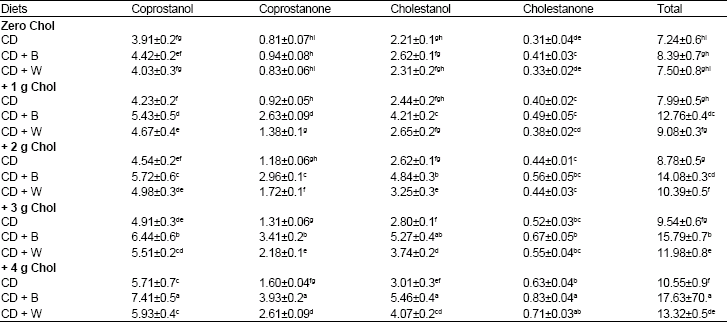 | |
CD: Control Diet. Values are means±SD. Means in a column without a common letter differ, p>0.05 | |
Triglycerides deposited as stored energy of the organism, while PL and cholesterol act as essential building blocks in the cell structures (Rotenberg and Jakobsen, 1978; Rolandelli et al., 1989). In rats fed barely and wheat the serum and liver lipids were significantly decreased compared to control rats and may due to the increased TG catabolism. As a result the ratio of PL/TG of serum was slightly increased from about (0.94) for rats fed control diet to about (0.98) for rats fed wheat and to about (1.1) for rats fed barely. Dietary fiber may be reduced the digestibility and absorption of lipids and resulted in low concentrations of blood lipids, the results are in agreement with the observations of Borel et al. (1990). Barely diet (rich in soluble fiber) resulted in significantly lower serum and liver lipids than wheat (rich in insoluble fiber) which indicated that soluble fiber had more lipid lowering effect than insoluble fiber. Dietary fat and cholesterol may escape intestinal absorption due to the increased viscosity of soluble fiber; bile acids may be bound to soluble fiber and removed from the enterohepatic circulation. Available dietary or synthesized cholesterol is normally converted to bile acids to help emulsify dietary fats in the digestive process. However, if cholesterol synthesis is suppressed by the presence of tocotrienol, a further shortage of bile acids may result, causing reduced fat emulsification and absorption.
| Table 8: | Bile acids in feces (mg/g dry wt) of rats fed cholesterol (Chol), barely (B) and wheat (W) for 28 days |
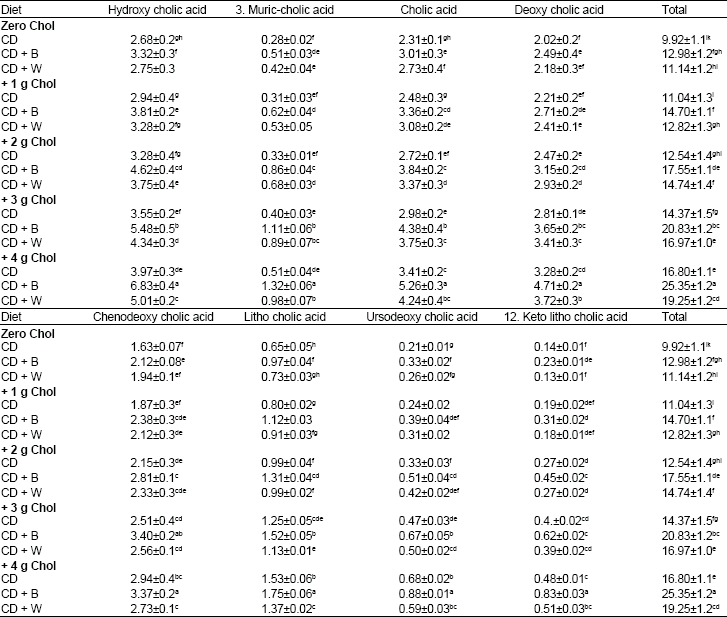 | |
CD: Control Diet. Values are means±SD. Means in a column without a common letter differ, p>0.05 | |
Several studies have reported a cholesterol-lowering effect with consumption of pulses, cereals as well as purified sources of soluble dietary fiber, including psyllium prepared from seed husks of Plantago ovata a native of India (Bell et al., 1989; Anderson and Siesei, 1990; Sola et al., 2007); oat (Avena sativa) products containing β-glucan (Shinnick et al., 1990; Davidson et al., 1991; Bridges et al., 1992) and guar gum the ground endosperm of Indian guar beans (Anderson and Siesei, 1990). Streppel et al. (2008) mentioned that every additional 10 g of recent dietary fiber intake per day reduced CHD mortality by 17%.
Most carefully controlled studies indicated that there are several mechanisms for the hypocholesterolemic effect of dietary fibers. Soluble fiber may bind bile acids or cholesterol in the intestine, preventing their reabsorption into the body. The liver responds by taking up more LDL-c from the blood stream thereby lowering the concentration of LDL-c in the blood. Short Chain Fatty Acids (SCFA) products of fermentation from soluble fiber in the gut, may inhibit synthesis of cholesterol by the liver, reducing the concentration of blood cholesterol. The high viscosity of soluble fiber may slow the rate of digestion and absorption of carbohydrates, affecting insulin activity, which is implicated in the removal of LDL-c in the blood (Andersson et al., 2002). Barely diet, fed to the rats caused significant reductions in the levels of TC in serum and endogenous organs, TG in serum and liver and LDL-c and HDL-c in serum compared with the wheat and basal diet. There was a significant decrease in the LDL-c/TC ratio and slight decrease in LDL-c/ HDL-c ratio in the serum of barely fed rats. Barely as oat contain soluble fiber and soluble β-glucan, which appeared to be strong predictors of the cholesterol-lowering in serum and liver of rats. The viscous property of soluble β-glucan may result in reduced absorption, or reabsorption of lipids (Kalra and Jood, 2000). Behall et al. (2004) observed that the addition of β-glucan from barley to the diet of hypercholesterolemia men and women lowered TC, LDL-c and TG concentrations and no significant difference between oats and barley was observed, which is an indication that β-glucan and not the source was critical in lipid reduction. Behall et al. (2004); Naumann et al. ( 2006); Keenan et al. (2007); Shimizu et al. (2008) showed that the barley and barley with high β-glucan content intake improves TC and LDL-c and significantly reduced visceral fat area during the trial among normal and dyslipidaemic subjects. In addition to the soluble fiber, barley contains ingredients, other than β-glucan, such as certain fatty acids, tocotrienols, saponins or phytosterols, suggested to play a role in the hypocholesterolemic effects of barley (Qureshi et al., 1986; Anderson and Siesei, 1990; Andersson, et al., 2002; McIntosh et al., 1991). It is therefore quite likely that barley has two distinct hypocholesterolemia constituents that are complementary in action.
Several studies have reported that the blood cholesterol concentration in rats responds to hypercholesterolemic diets in a manner similar to that seen in humans fed high saturated fat and high cholesterol diets (Lichtenstein et al., 1994; Schnohr et al., 1994; Ginsberg et al., 1995). Thus rats were used in the present study to investigate the hypercholesterolemic effects of different levels of dietary cholesterol which resulted in significantly increase in serum and liver TC, serum LDL-c and fecal neutral sterols and bile acids. Observed results after cholesterol ingestion were similar to those responses reported in rats (Mokady and Liener, 1982), hamsters (Ranhotra et al., 1998), chicks (Castillo et al., 1999) and humans (Mistry et al., 1981). There are two major pathways that mammalian cells use to supply themselves with cholesterol, one involving the synthesis of sterols from acetyl-CoA and the other the metabolism of cholesterol-rich lipoprotein particles via receptor-mediated endocytosis. There also are several pathways that mammalian cells use to break down cholesterol, and these disposal pathways are equal in physiological importance to the supply pathways. A major catabolic route involves conversion of cholesterol into bile acids, a transformation mediated by liver enzymes (Russell, 2009).
Barely diet resulted in significantly more fecal neutral sterols and bile acids than wheat and control diet. This significant role of barely compared with wheat was also showed after addition cholesterol to the rat diets. Although wheat bran increases fecal weight, it does not usually increase fecal bile acid excretion (Kay and Truswell, 1977; Lampe et al., 1991). Furthermore, wheat bran increases SCFA production in the colon to a lesser extent than does soluble fiber (Bridges et al., 1992). Soluble fibers increase fecal bile acid excretion and SCFA production in the colon to a greater extent than do wheat bran and other insoluble fibers. Diets containing more soluble fibers such as β-glucans affected the excretion of bile acids and neutral sterols the most. These results suggest that dietary fiber-rich barley-containing diets have beneficial physiologic effects (Dongowski et al., 2002). This study confirms prior studies in rats showing that dietary fiber, especially soluble fiber significantly decrease serum and liver lipid concentrations (Jennings et al., 1988; Shinnick et al., 1990). Lower serum and liver lipid values of rats fed barley compared with wheat and control diets were also in agreement with earlier reports (Newman et al., 1990). One possible interpretation of this phenomenon is that cholesterol is withdrawn from the liver to replace dietary cholesterol and/or bile acids being removed from the digestive tract by the soluble fiber components (Martinez et al., 1992). The higher excreta crude fat content of barley fed rats suggests that the reduction in plasma cholesterol is also due to lessened absorption of dietary fat. The viscosity associated with high soluble fiber barleys is postulated to interfere with fat absorption in the intestine (Vahouny and Cassidy, 1985; Kalra and Jood, 2000; Dongowski et al., 2002). The high concentration of cholesterol in serum and livers of rats fed wheat compared with rats fed barley can be compared with work by Shinnick et al. (1990) wherein rats fed 1% cholesterol had elevated serum and liver cholesterol concentrations, which were decreased by 25 and 39%, respectively, by 10% dietary fiber in the form of oat bran flour.
Conclusion: In conclusion, the addition of barley to a healthy diet may be effective in improving human lipid profile. In terms of the public health significance of these findings, it is practical to achieve a significant increase in dietary fiber when more cereal-based foods are included. Barely is capable of lowering plasma cholesterol concentrations when substituted for the more commonly ingested wheat-based foods. There are a variety of foods that can be prepared from suitable strains of appropriately milled barley and modern technology offers the means of overcoming any limitations in palatability or acceptability of such foods.