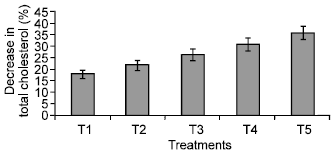
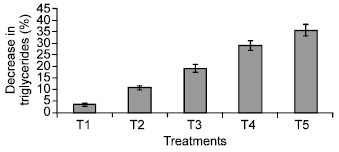
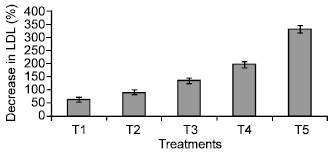
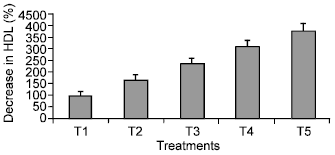
Research Article
Extraction of ß-glucan from Oat and its Interaction with Glucose and Lipoprotein Profile
Department of Food Science and Technology, Pir Mehr Ali Shah Arid Agriculture University, Rawalpindi, Pakistan
Faqir Muhammad Anjum
National Institute of Food Science and Technology, University of Agriculture Faisalabad, Pakistan
Tahir Zahoor
National Institute of Food Science and Technology, University of Agriculture Faisalabad, Pakistan
Haq Nawaz
Institute of Animal Nutrition and Feed Technology, University of Agriculture Faisalabad, Pakistan