Research Article
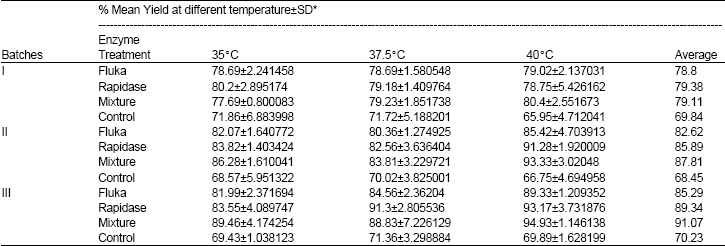
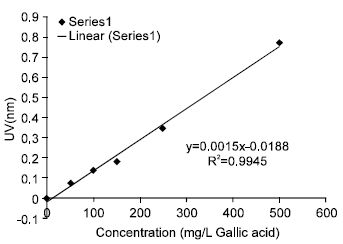
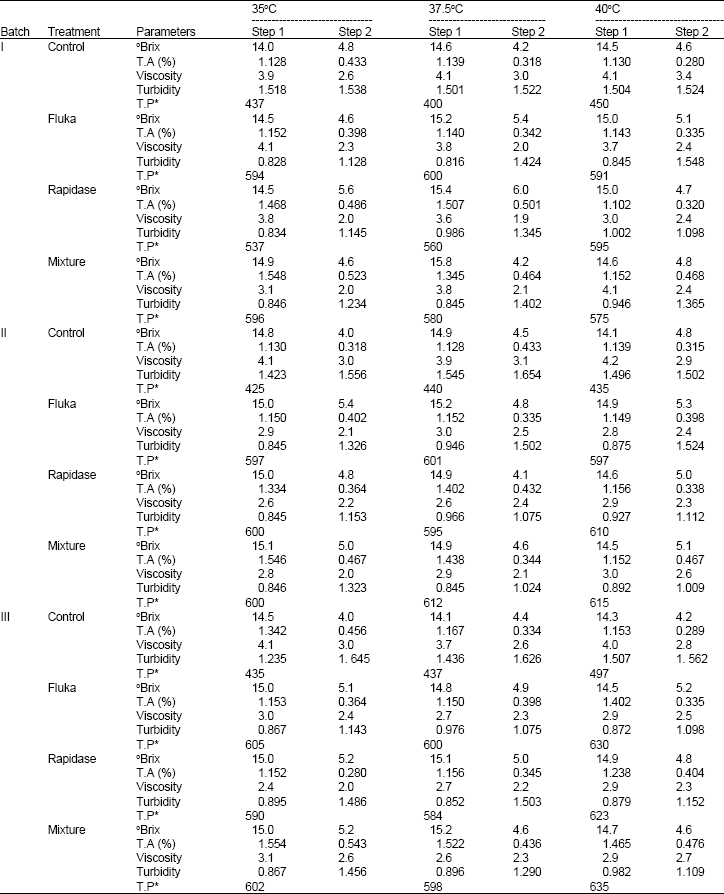
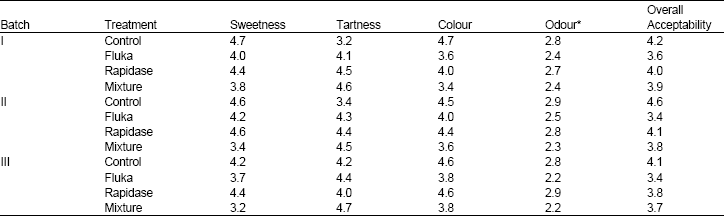
The Influence of a Pectinase and Pectinase/hemicellulases Enzyme Preparations On percentage Pineapple Juice Recovery, Particulates and Sensory Attributes
Department of Food Science and Engineering, School of Food Science and Technology,Jiangnan University, Wuxi 214122, PR China
Zhang Wang
Pwani University College, P.O. Box 195, Kilifi, Kenya, East Africa
Shi-Ying Xu
Department of Food Science and Engineering, School of Food Science and Technology,Jiangnan University, Wuxi 214122, PR China
Wenbin Zhang
Department of Food Science and Engineering, School of Food Science and Technology,Jiangnan University, Wuxi 214122, PR China