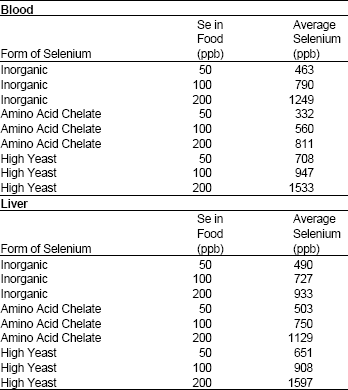
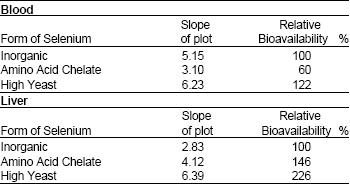
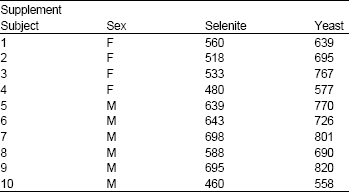
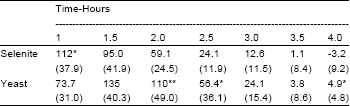
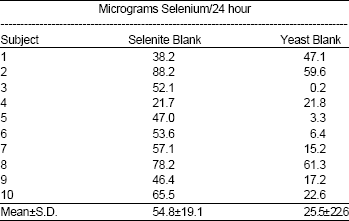
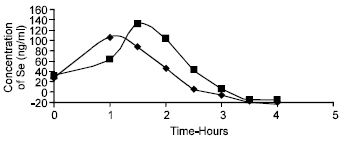
Research Article
The Relative Bioavailability of Sodium Selenite and High Selenium Yeast in Human
Department of Medical Allied Sciences, Al-Zarka University College, Al-Balqa Applied University, Jordan
Monzer M. Krishan
Department of Medical Allied Sciences, Al-Zarka University College, Al-Balqa Applied University, Jordan
Ramadan K. Hejazin
Department of Medical Allied Sciences, Al-Zarka University College, Al-Balqa Applied University, Jordan
Ayman S. Mazahreh
Department of Applied Sciences, Princess Alia University College, Al-Balqa Applied University, Jordan
Mohammed Al - Damanhoory
Department of Applied Sciences, Princess Alia University College, Al-Balqa Applied University, Jordan
Jihad M. Quasem
Department of Medical Allied Sciences, Al-Zarka University College, Al-Balqa Applied University, Jordan