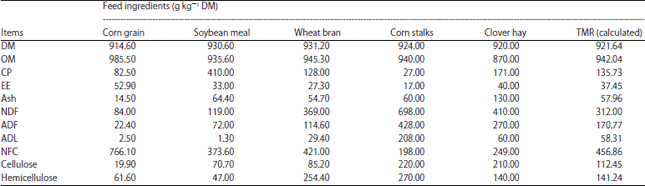
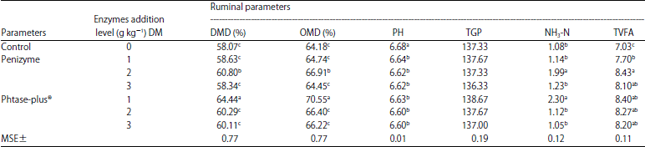
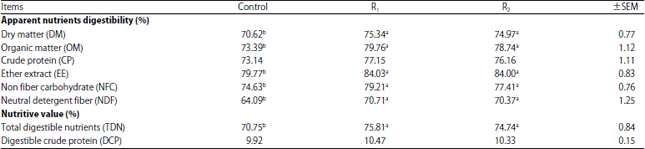
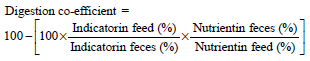Research Article
Effect of Xylanase and Phytase Supplementation on Goat’s Performance in Early Lactation
Department of Dairy Sciences, National Research Centre, 33 Buhouth St, Dokki, Giza, Egypt
LiveDNA: 20.981
A.A. Aboamer
Department of Dairy Sciences, National Research Centre, 33 Buhouth St, Dokki, Giza, Egypt
Hoda Alzahar
Department of Dairy Sciences, National Research Centre, 33 Buhouth St, Dokki, Giza, Egypt
M.M. Abdo
Department of Dairy Sciences, National Research Centre, 33 Buhouth St, Dokki, Giza, Egypt
H.A. Murad
Department of Dairy Sciences, National Research Centre, 33 Buhouth St, Dokki, Giza, Egypt
LiveDNA: 20.630