Research Article
Acute and Cytotoxicity Studies of Aqueous and Ethanolic Leaf Extracts of Chromolaena odorata
Department of Industrial Chemistry, Nnamdi Azikiwe University, Awka, Nigeria
A.N. Ezejiofor
Department of Experimental Pharmacology and Clinical Pharmacy,University of Port Harcourt, Port Harcourt, Nigeria
P.N. Okafor
Department of Experimental Pharmacology and Clinical Pharmacy,University of Port Harcourt, Port Harcourt, Nigeria
I.I. Ijeh
Department of Experimental Pharmacology and Clinical Pharmacy,University of Port Harcourt, Port Harcourt, Nigeria










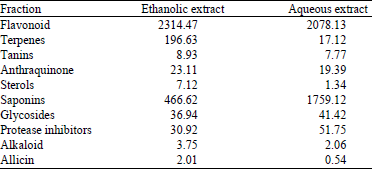
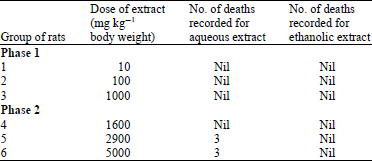
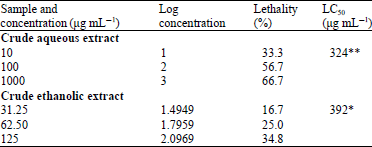
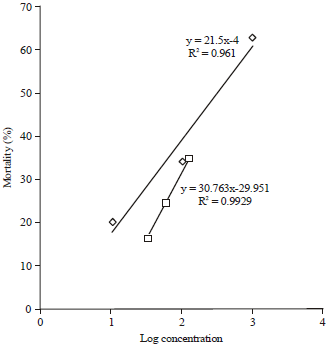
Nguyen Tam Reply
Hello
I am a chemistry teacher in Vietnam
I have read through the article about your research.
"Acute and Cytotoxicity Studies of Aqueous Extracts of Chromolaena and Ethanolic Leaf odorata"
It was great, I have found a lot of information from it.
If you do not mind, could you please give me more information about your research. I need to refer it to guide a group of students to do scientific research.
I think the results from this study will help us find some valuable medicinal products.
Thank you very much!
(Forgive me for using "google translate" to write to you)