Clinical Report
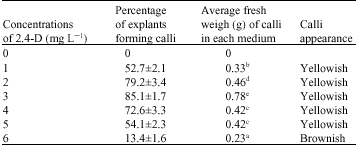
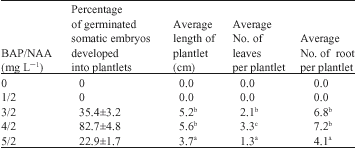
In vitro Regeneration of Irvingia gabonensis by Somatic Embryogenesis
Laboratory of Biochemical and Plant Physiology, Department of Biological Sciences, Higher Teacher Training School, P.O. Box 47, Yaounde, Cameroon
Oumar
Jay P. J. Biotechnology Laboratory, Institute of Agricultural Research for Development (IRAD), P.O. Box 25, Buea, Ekona, Cameroon
Niemenak Nicolas
Laboratory of Biochemical and Plant Physiology, Department of Biological Sciences, Higher Teacher Training School, P.O. Box 47, Yaounde, Cameroon
Donfagsiteli Tchinda Nehemie
Laboratory of Botanic and Traditional Medicine, Institute of Medical Research and Medicinal Plants Studies (IMPM), P.O. Box 6163, Yaounde, Cameroon
Omokolo Ndoumou Denis
Laboratory of Biochemical and Plant Physiology, Department of Biological Sciences, Higher Teacher Training School, P.O. Box 47, Yaounde, Cameroon