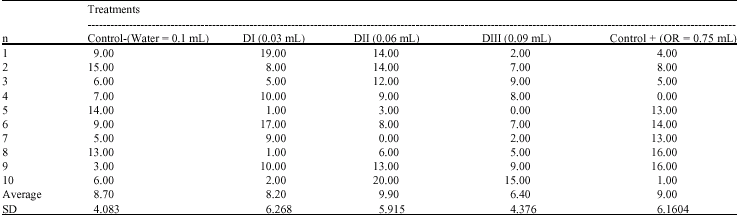
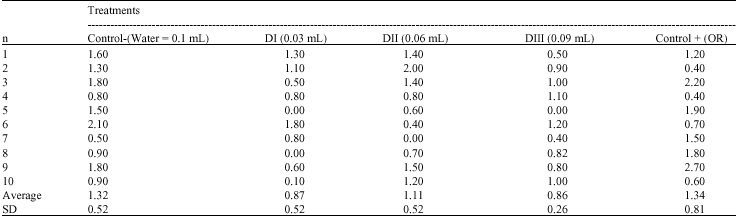
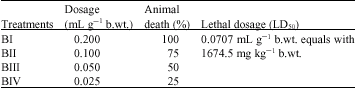
Research Article
Characteristics, Efficacy and Safety Testing of Standardized Extract of Croton tiglium Seed from Indonesia as Laxative Material
Department of Agroindustrial Technology, Faculty of Agricultural Technology, Bogor Agricultural University, Bogor, 16002, Indonesia
Djumali Mangunwidjaja
Department of Agroindustrial Technology, Faculty of Agricultural Technology, Bogor Agricultural University, Bogor, 16002, Indonesia
Sapta Raharja
Department of Agroindustrial Technology, Faculty of Agricultural Technology, Bogor Agricultural University, Bogor, 16002, Indonesia
Leonardus B.S. Kardono
Research Center for Chemistry, Indonesian Institute of Sciences, Kawasan PUSPIPTEK Serpong, 15310, Indonesia
Dyah Iswantini
Department of Chemistry, Faculty of Mathematics and Natural Sciences, Bogor Agricultural University, Bogor, Indonesia