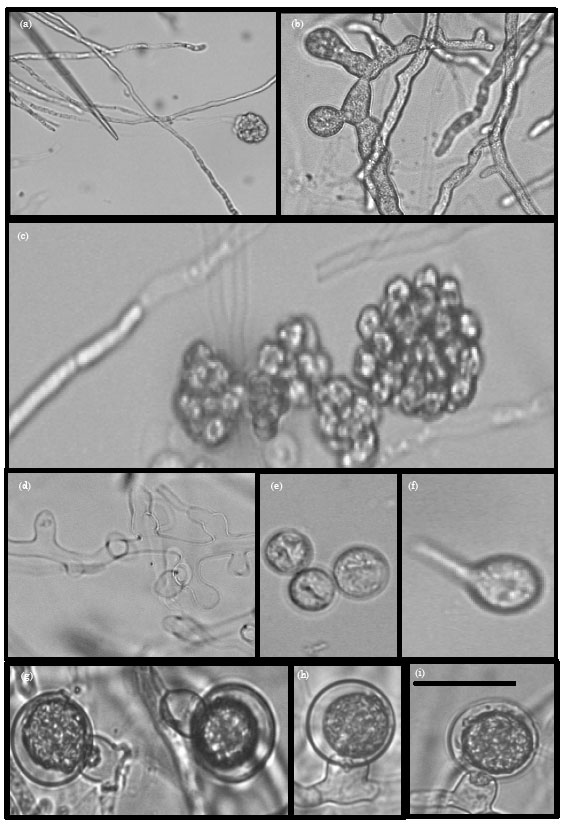
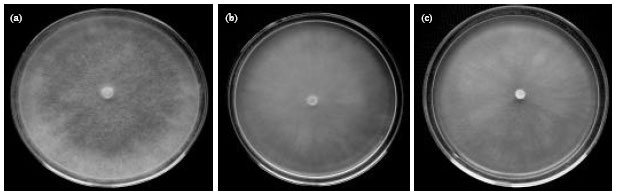
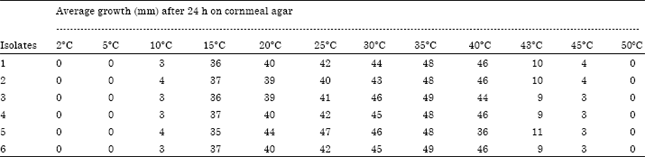
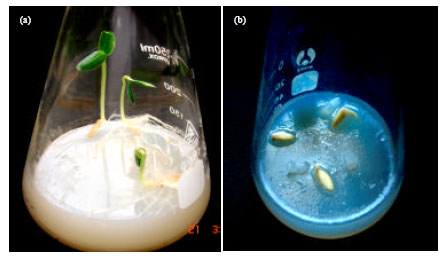
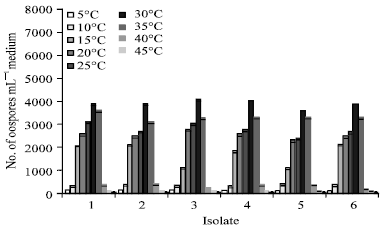
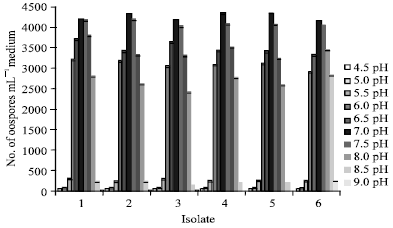
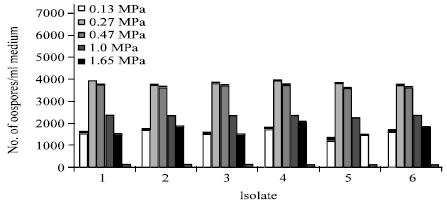
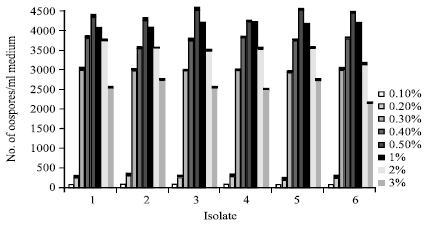
Research Article
Materials for Pythium Flora of Saudi Arabia (I) Occurrence, Pathogenicity and Physiology of Reproduction of Pythium aphanidermatum (Edson) Fitzp. Isolated from North and East Regions of Saudi Arabia
Department of Biology, College of Science, King Faisal University, Al-Hassa, Saudi Arabia
Hani M.A. Abdelzaher
Department of Biology, College of Sciences, Al-Jouf University, Sakaka, Saudi Arabia