Research Article
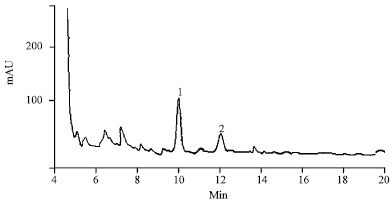
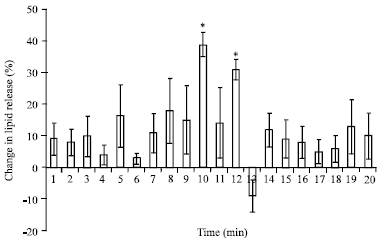
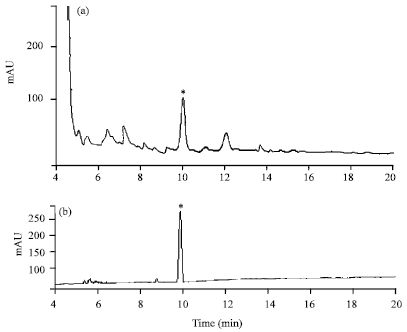
Identification of an AKH/RPCH Family Peptide in the Rice Grasshopper, Hieroglyphus banian (Acrididae: Orthoptera)
Division of Insect Biochemistry and Physiology, Department of Zoology, University of Calicut, Kerala, 673635, India
A.P. Ajaykumar
Division of Insect Biochemistry and Physiology, Department of Zoology, University of Calicut, Kerala, 673635, India
D. Umadevi
Division of Insect Biochemistry and Physiology, Department of Zoology, University of Calicut, Kerala, 673635, India
V.S. Binitha
Division of Insect Biochemistry and Physiology, Department of Zoology, University of Calicut, Kerala, 673635, India
K.M. Ismail
Division of Insect Biochemistry and Physiology, Department of Zoology, University of Calicut, Kerala, 673635, India
K.A. Rasheed
Division of Insect Biochemistry and Physiology, Department of Zoology, University of Calicut, Kerala, 673635, India