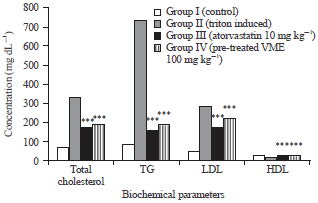
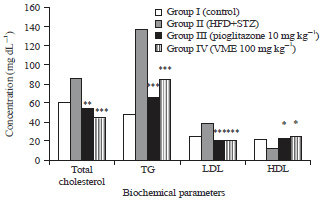
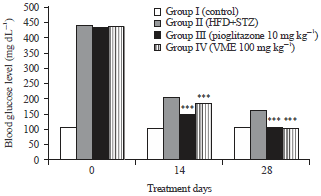
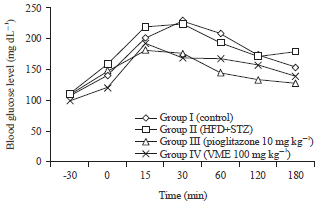
Research Article
Preventive and Curative Potential of Vigna mungo against Metabolic Syndrome in Acute and Chronic Rat Models
SPP School of Pharmacy and Technology Management, SVKM�s NMIMS, V.L. Mehta Road, Vile Parle (W), Mumbai, 400056, Maharashtra, India
Rachana Somaiya
SPP School of Pharmacy and Technology Management, SVKM�s NMIMS, V.L. Mehta Road, Vile Parle (W), Mumbai, 400056, Maharashtra, India
Sameer Patel
SPP School of Pharmacy and Technology Management, SVKM�s NMIMS, V.L. Mehta Road, Vile Parle (W), Mumbai, 400056, Maharashtra, India