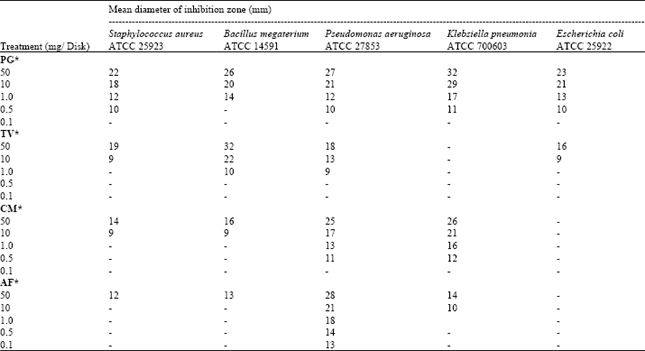
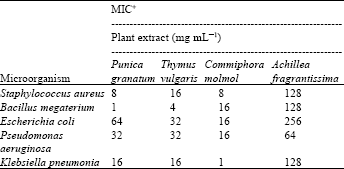
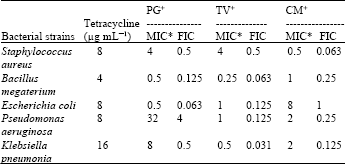
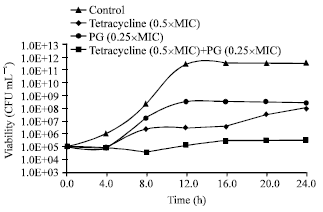
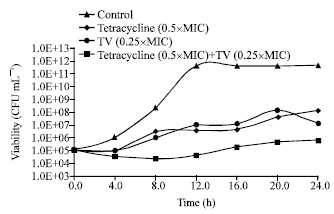
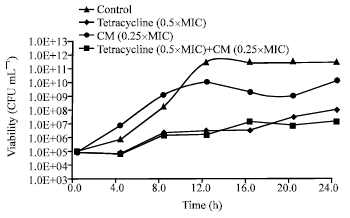
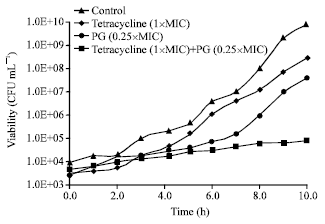
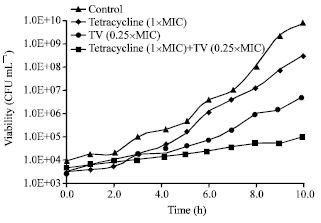
Research Article
Synergic Interactions Between Selected Botanical Extracts and Tetracycline Against Gram Positive and Gram Negative Bacteria
Department of Biological Sciences, Faculty of Science, King Faisal University, Al-Hufof 31982, Ahsaa, KSA
W.M. El-Sayed
Department of Biological Sciences, Faculty of Science, King Faisal University, Al-Hufof 31982, Ahsaa, KSA