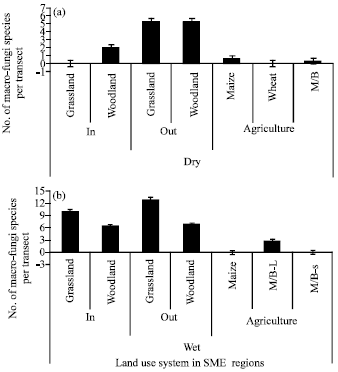
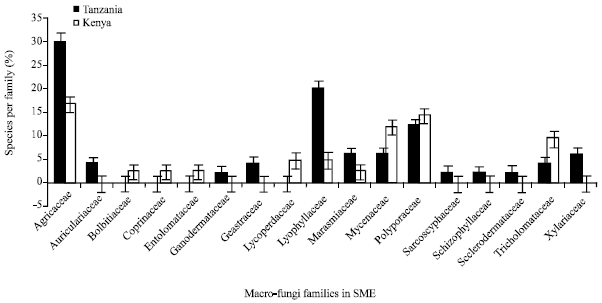
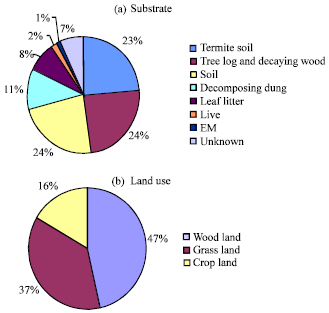
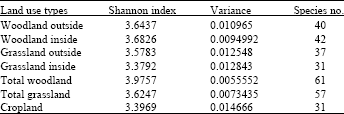
Research Article
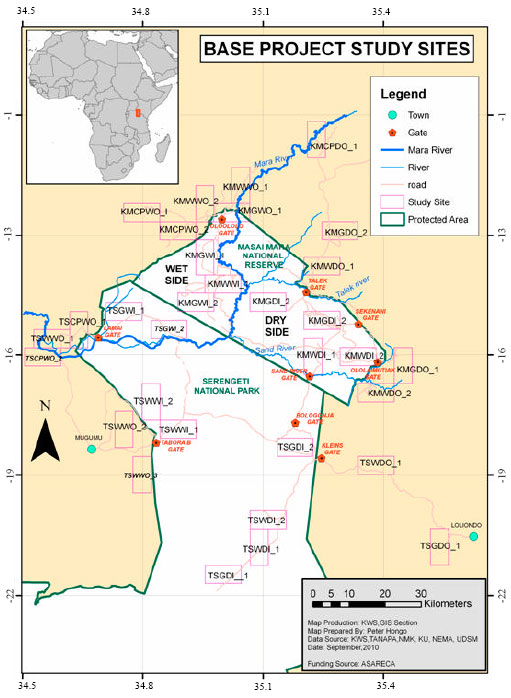
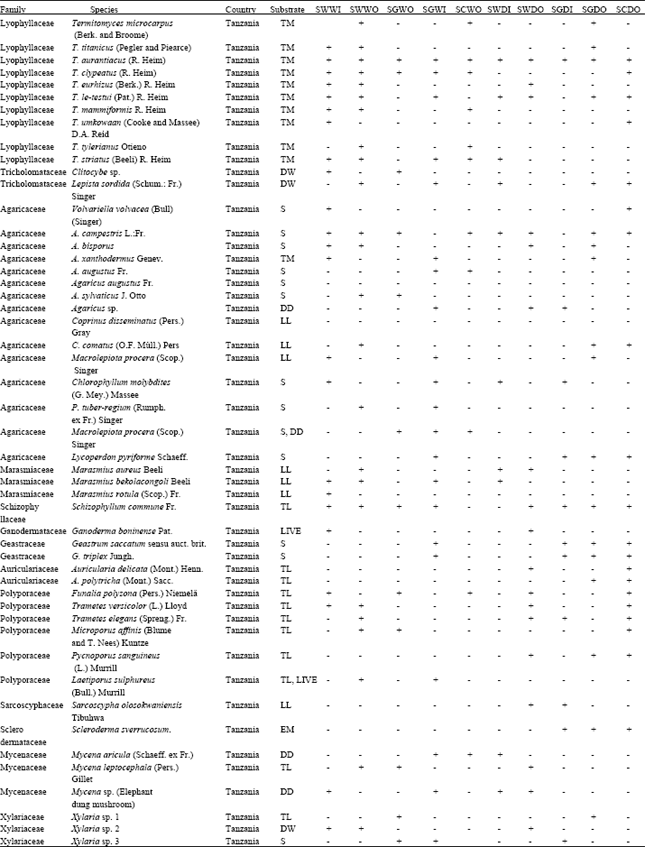
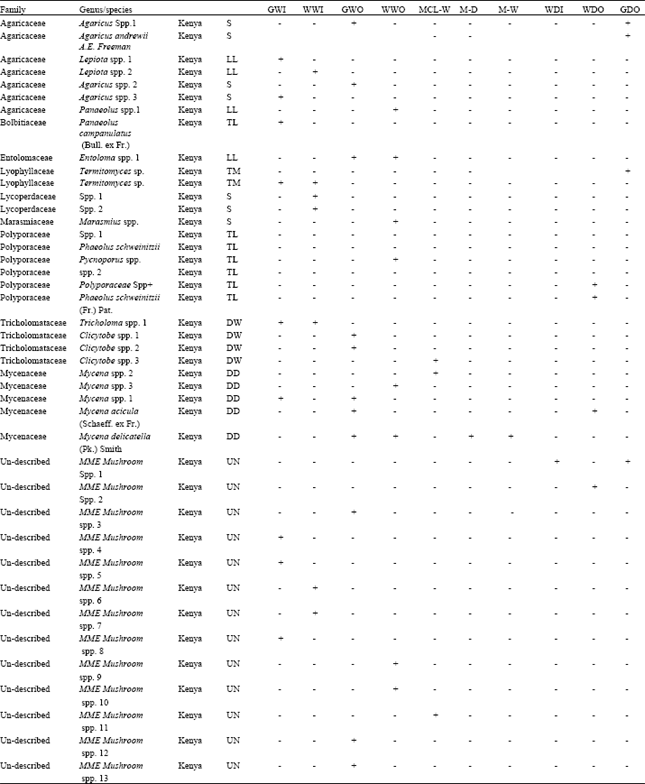
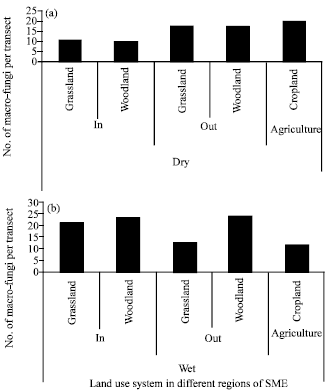
An Inventory of Macro-fungi and their Diversity in the Serengeti-Masai Mara Ecosystem, Tanzania and Kenya
Department of Molecular Biology and Biotechnology, University of Dar es Salaam, P.O. Box 35179, Dar es Salaam, Tanzania
M.N. Muchane
Department of Botany and Zoology, National Museums of Kenya, P.O. Box 40658, Nairobi, Kenya
C.W. Masiga
ASARECA P.O. Box 765, Entebbe, Uganda
C. Mugoya
ASARECA P.O. Box 765, Entebbe, Uganda
M. Muchai
Department of Botany and Zoology, National Museums of Kenya, P.O. Box 40658, Nairobi, Kenya