Research Article
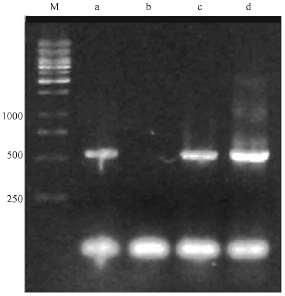
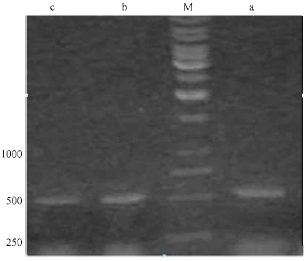
Identification of an Isolate of Pseudomonas aeroginosa Deposited in PTCC as a PHA Producer Strain: Comparison of Three Different Bacterial Genomic DNA Extraction Methods
Department of Pharmaceutical Biotechnology and Isfahan Pharmaceutical Research Center, School of Pharmacy, Isfahan University of Medical Sciences and Health Services, Isfahan, Iran
Ali Jahanian Najafabadi
Department of Pharmaceutical Biotechnology and Isfahan Pharmaceutical Research Center, School of Pharmacy, Isfahan University of Medical Sciences and Health Services, Isfahan, Iran
Daryoush Abedi
Department of Pharmaceutical Biotechnology and Isfahan Pharmaceutical Research Center, School of Pharmacy, Isfahan University of Medical Sciences and Health Services, Isfahan, Iran
Abbas Jafarian Dehkordi
Department of Pharmaceutical Biotechnology and Isfahan Pharmaceutical Research Center, School of Pharmacy, Isfahan University of Medical Sciences and Health Services, Isfahan, Iran