Research Article
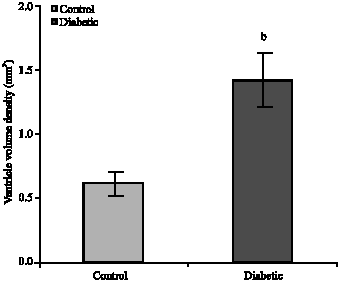
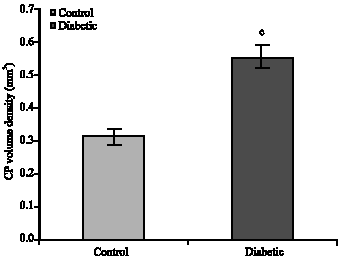
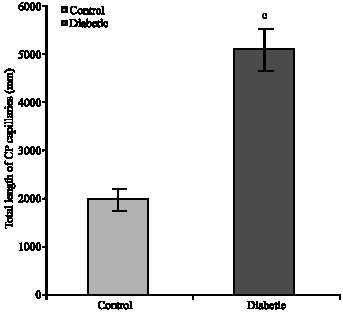
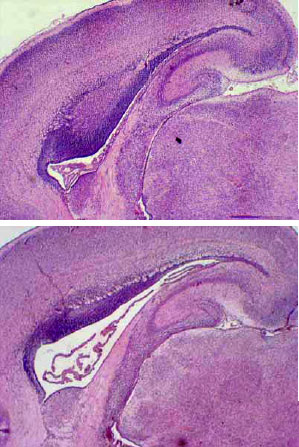
Maternal Hyperglycemia Proliferate Choroids Plexus and Enlarge the Lateral Ventricle in Brain of Newborn Rats
Department of Biology, Faculty of Science, Islamic Azad University of Mashhad, Iran
Behnam-Rassouli Morteza
Department of Biology, Faculty of Science, Islamic Azad University of Mashhad, Iran
Rahimi Amin
Department of Biology, Faculty of Science, Islamic Azad University of Mashhad, Iran