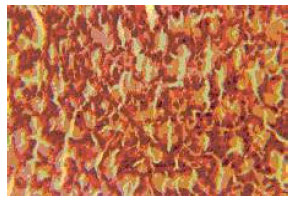
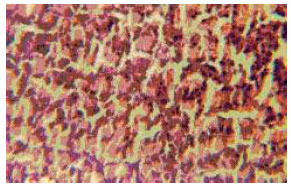
Research Article
Hypoglycemic Effect of Ficus microcarpa Leaves (Chinese Banyan) on Alloxan-induced Diabetic Rats
Department of Pharmacology, College of Pharmacy, Sri Ramakrishna Institute of Paramedical Sciences,395, Sarojini Naidu Road, Coimbatore-641 044, India
M. Uma Maheswari
Department of Pharmacology, College of Pharmacy, Sri Ramakrishna Institute of Paramedical Sciences,395, Sarojini Naidu Road, Coimbatore-641 044, India
A.T. Sivashanmugam
Department of Pharmacology, College of Pharmacy, Sri Ramakrishna Institute of Paramedical Sciences,395, Sarojini Naidu Road, Coimbatore-641 044, India
V. Subhadra Devi
Department of Pharmacology, College of Pharmacy, Sri Ramakrishna Institute of Paramedical Sciences,395, Sarojini Naidu Road, Coimbatore-641 044, India
N. V. Prasanth
Department of Pharmacology, College of Pharmacy, Sri Ramakrishna Institute of Paramedical Sciences,395, Sarojini Naidu Road, Coimbatore-641 044, India
T.K. Ravi
Department of Pharmacology, College of Pharmacy, Sri Ramakrishna Institute of Paramedical Sciences,395, Sarojini Naidu Road, Coimbatore-641 044, India