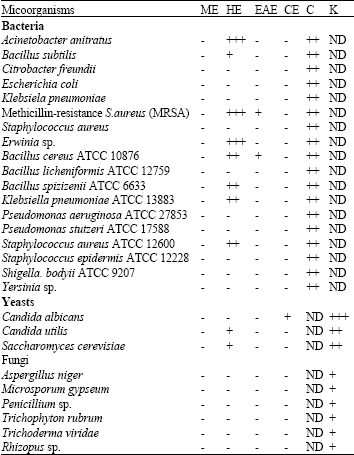
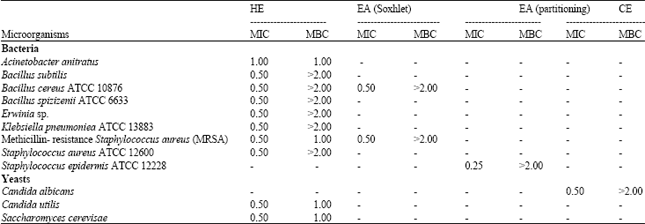
Research Article
Antimicrobial Activity of Various Extracts of a Tropical Chlorophyta Macroalgae, Halimeda discoidea
School of Biological Sciences, Universiti Sains Malaysia, Penang, Malaysia
I. Darah
School of Biological Sciences, Universiti Sains Malaysia, Penang, Malaysia
S. Shaida Fariza
School of Biological Sciences, Universiti Sains Malaysia, Penang, Malaysia
M.K. Mohd Jain Nordin
School of Chemical Sciences, Universiti Sains Malaysia, Penang, Malaysia
Z. Nurul Aili
School of Biological Sciences, Universiti Sains Malaysia, Penang, Malaysia