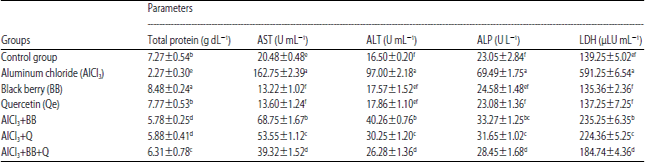
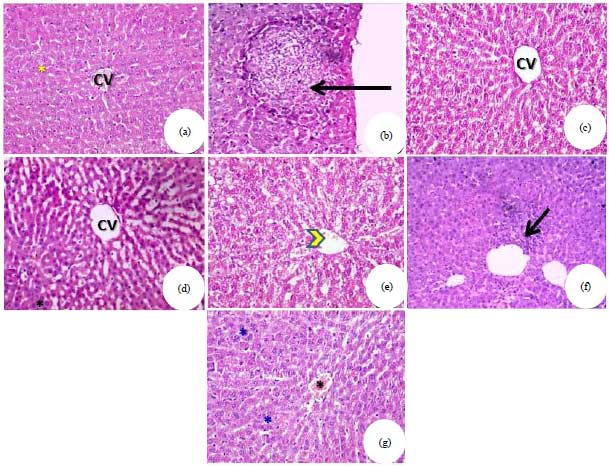
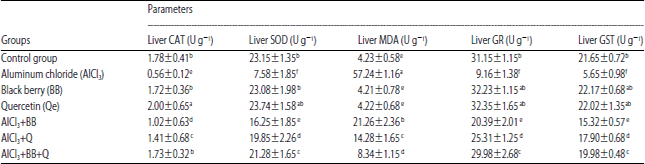
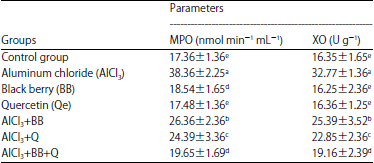
Research Article
Antioxidant, Protective Effect of Black Berry and Quercetin Against Hepatotoxicity Induced by Aluminum Chloride in Male Rats
Department of Biology, Faculty of Science, Taif University, 888 Taif, Kingdom of Saudi Arabia
LiveDNA: 966.26710