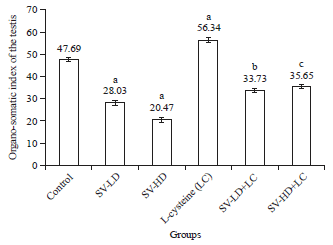
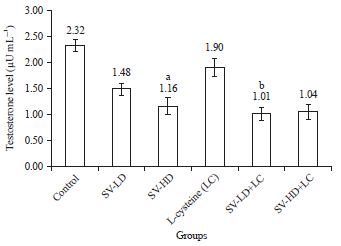
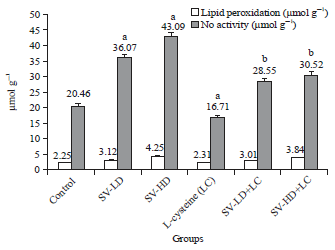
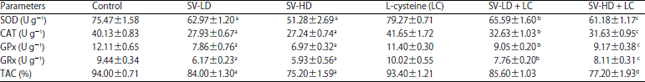
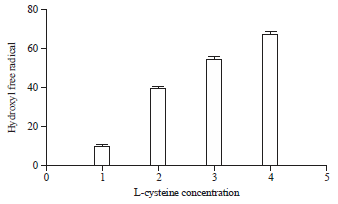
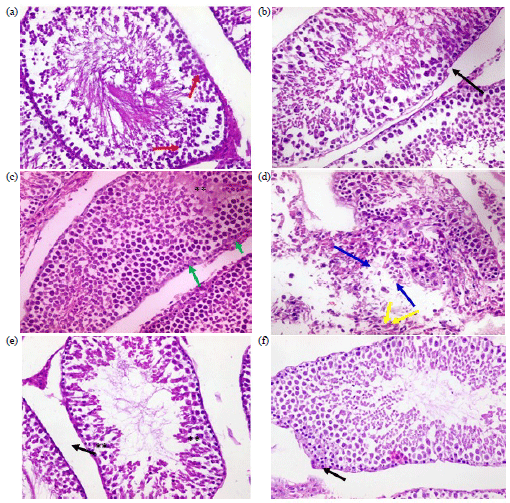
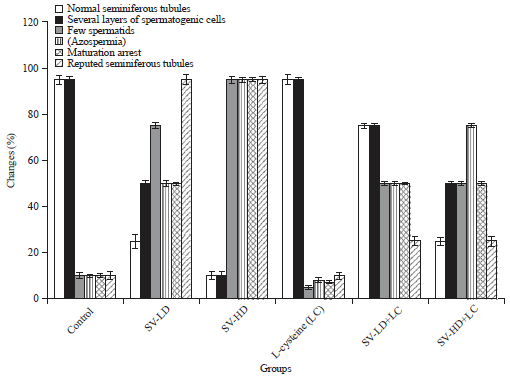
Research Article
Protective Effect of L-cysteine Against Sodium Valproate-induced Oxidant Injury in Testis of Rats
Department of Biology, Faculty of Science, Taif University 888, Taif, Saudi Arabia
Rasha Al-Eisa
Department of Biology, Faculty of Science, Taif University 888, Taif, Saudi Arabia
Reham Z. Hamza
Department of Biology, Faculty of Science, Taif University 888, Taif, Saudi Arabia
Howayda E. Khaled
Department of Zoology, Faculty of Science, Suez University, Suez, Egypt
Nahla S. El-Shenawy
Department of Zoology , Faculty of Science, Suez Canal University, Ismailia, 41522, Egypt
LiveDNA: 20.782