Research Article
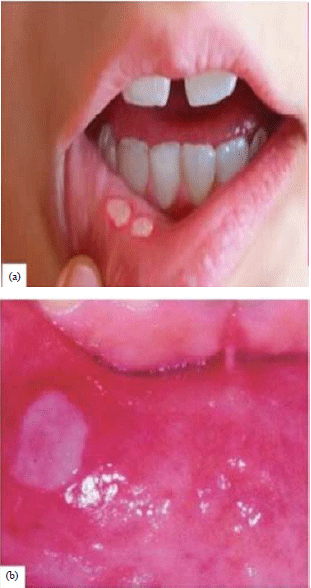
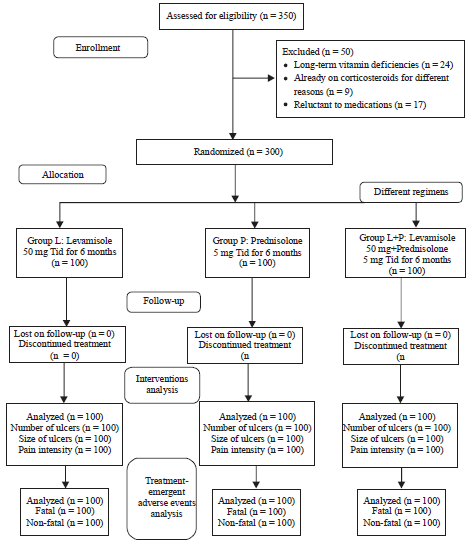
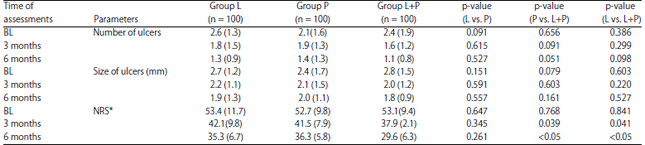
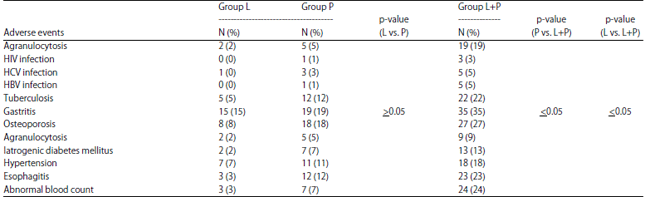
A Comparison of Immunomodulatory Monotherapy and Combination Therapy for Recurrent Aphthous Stomatitis
Department of Stomatology, PLA Army General Hospital, 100700 Beijing, China
Jiyan Liu
Department of Stomatology, PLA Army General Hospital, 100700 Beijing, China
Wenfeng Zhao
Department of Stomatology, PLA Army General Hospital, 100700 Beijing, China
LiveDNA: 86.16516