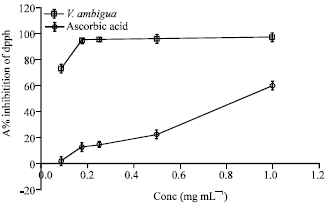
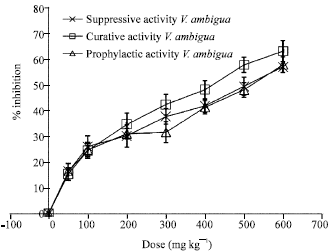
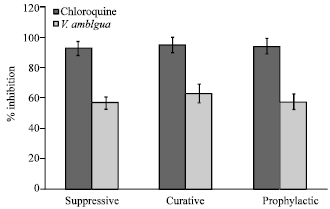
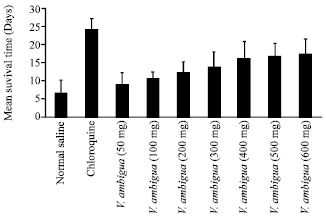
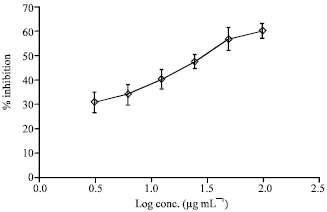
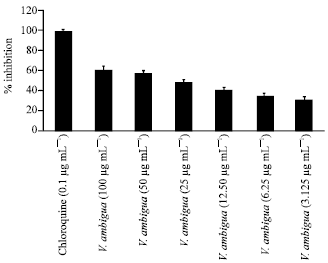
Research Article
Evaluation of the Antimalarial Potential of Vernonia ambigua Kotschy and Peyr (Asteraceae)
Department of Pharmacology, College of Medical Sciences, Bingham University, Jos, Nigeria
N. N. Wannang
Department of Pharmacology, Faculty of Pharmaceutical Sciences, University of Jos, Nigeria
G. A. Ajoku
Department of Microbiology and Biotechnology, National Institute for Pharmaceutical Research and Developmen Idu, Abuja, Nigeria
P. F. Builders
Department of Pharmaceutical Technology and Raw Materials Development, National Institute for Pharmaceutical Research and Developmen Idu, Abuja, Nigeria
A. Orisadipe
Department of Medicinal Plant Research and Traditional medicine, National Institute for Pharmaceutical Research and Developmen Idu, Abuja, Nigeria
J. C. Aguiyi
Department of Pharmacology, Faculty of Pharmaceutical Sciences, University of Jos, Nigeria