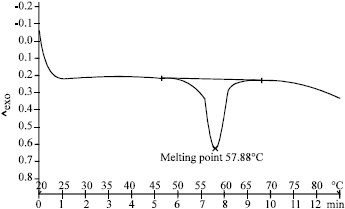
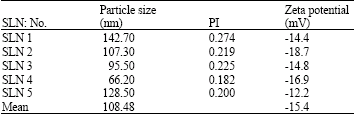
Research Article
Solid Lipid Nanoparticles Preparation and Characterization
Laboratory Immunotherapeutic and Vaccine, Institute of Bioscience, Universiti Putra Malaysia, Serdang 43400, SDE, Malaysia
A. Rasedee
Department of Pathology and Microbiology, Faculty of Veterinary Medicine, Universiti Putra Malaysia, Serdang 43400, SDE, Malaysia