Research Article
Effect of Administration of Fungal Mycotoxin (Gliotoxin) on Clinical and Serobiochemical Parameters in Camels
Department of Microbiology, King Faisal University, P.O. Box 1757, Al-Ahsa 31982, Kingdom of Saudi Arabia
Gliotoxin is a mycotoxin produced by a number of fungi including important human and animal pathogens as Aspergillus fumigatus (Bauer, 1994; Eichner et al., 1988). Gliotoxin affects the rumen fermentation, reducing digestibility and volatile fatty acid production in vitro (Morgavi et al., 2004). Reeves et al. (2004) using an insect model demonstrated the significance of gliotoxin in increasing the virulence of A. fumigatus. Gliotoxin is a well studied non-ribosomal peptide toxin (Gardiner and Howlett, 2005) and has long been fingered as a putative factor contributing to invasive aspergillosis due to its cytotoxic (Grovel et al., 2002), genotoxic (Nieminen et al., 2002) and apoptotic properties (Hesseltine, 1986; Kweon et al., 2003; Niyo et al., 1988; Pahl et al., 1996; Schiefer, 1990). Presently, the difficulty of diagnosis increased due to limited research, occurrence of multiple mycotoxins, non-uniform distribution, interactions with other factors and problems of sampling and analysis. Because of the difficulty of diagnosis, the determination of a mycotoxin problem becomes a process of elimination and association. This study was designed to investigate the clinical and serobiochemical effects induced by gliotoxin in camels in an effort to diagnose the fungal infection in camels.
Ten healthy and mature (3-4 years old having 200-250 kg b.wt.) camels were used in the study. They were kept in an open yard with free access to food and water. Animals were divided randomly into two groups. Group A animals were treated with saline and kept as controls. Group B animals were injected with gliotoxin (Sigma Chemicals, UK). In a pilot experiment, the dose of 0.2 μg kg-1 b.wt., administered intravenously (IV) killed all the three animals tested. Therefore, a bolus of 0.1 μg kg-1 b.wt. was administered IV to group B animals. The blood samples were collected daily for seven days into plain tubes. Serum was separated and stored at -30°C until analysis.
Serum biochemical analysis: The serum was analyzed for total protein, urea nitrogen, creatinine, glucose, total bilirubin, aspartate amino-transferase (AST), Gamma Glutamyl Transferase (GGT), alkaline phosphatase (ALKP), Sorbitol Dehydrogenase (SD) and Lactic Dehydrogenase (LD) by automated methods (Hitachi 704 Automatic Analyzer, Hitachi, Tokyo, Japan). Anion gap was quantified and calculated as = [Na] + [K]-([Cl] + [Hco3]) (Constable et al., 1997).
RESULTS
The administration of gliotoxin to camels produced anorexia and the animals were depressed and lethargic after day 4 of administration. The serobiochemical profiles of camels administered with gliotoxin are shown in Table 1. The sampling day and treatment interactions were detected (p<0.01) from day 2 of administration for all parameters except creatinine, protein, urea nitrogen and lactic dehydrogenase.
| Table 1: | Effect of administration of gliotoxin (0.1 μg kg-1) on some serobiochemical parameters in camels |
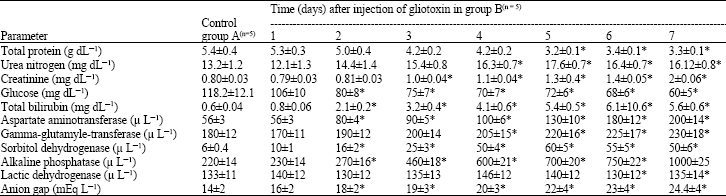 | |
| *p<0.01, significantly different from controls | |
The effect of gliotoxin administration was observed on day 3 for creatinine, on day 4 for urea nitrogen and on day 5 for protein. However, no effect was observed on the activity of LD. Higher concentration levels of urea nitrogen, Creatinine, total bilirubin and anion group (p<0.01) were observed in group B as compared to group A animals. Also, increased activity (p<0.01) of ASP, GGT, SD and ALKP was found in group B than group A animals. A decrease (p<0.01) in concentration of protein and glucose was observed in group B than group A animals.
It was found in this study that the camels administered with gliotoxin were lethargic and anorexic. They were also depressed from day 3 until day 7. The gliotoxin has been proved to be toxic to a number of animal species in vivo (Eichner et al., 1988; Bauer, 1994) and in vitro (Morgavi et al., 2004). The serum protein concentration decreased on day 5 after administration but the serum glucose concentration decreased at a faster rate in gliotoxin treated animals. This effect might be due to decrease in appetite and suggests that the treated animals were in a state of negative energy balance. Animals treated with gliotoxin maintained lower serum glucose even in the pre-diabetic state (Larsen et al., 2000). However, animals treated chronically with small doses of gliotoxin showed an increase in weight gains as compared to those in controls (Larsen et al., 2000). An increase in serum bilirubin in animals treated with gliotoxin suggested hepatic dysfunction (Cornelius, 1987). Serum activities of AST, GGT, SD and ALKP were also higher in these animals thus reflecting damage to the liver. The AST is present in high concentration in muscle, liver and brain and the serum elevation indicates damage to one or more of their tissues (Murray et al., 1990). Elevation of GGT, SD and ALKP activities are indices of damaged liver and obstruction of the biliary tract (Cornelius, 1989). Similar increases in liver enzymes were reported in calves (Osweiler et al., 1993), lambs (Edrington et al., 1995), swine (Colvin et al., 1993), horses (Ross et al., 1993) and broiler chicks (Ledoux et al., 1992) intoxicated with another mycotoxin fumonisin. The concentration of both the urea nitrogen and creatinine in serum increased in gliotoxin treated animals. Elevations of both metabolites were observed in cases of acute renal insufficiency and urinary tract obstruction (Murray et al., 1990). However, a few researchers have reported an increase in these indices in case of decreased plasma volume, renal blood flow and glomerular blood flow (Smith et al., 1999; Constable et al., 2000). The anion gap also increased in treated camels from day 3 onward reflecting metabolic acidosis (Mathur et al., 2001). Such effects may also be due to the presence of anions associated with uremia (Constable, 1997).
The gliotoxin administrated IV at a dose of 0.1 μg kg-1 b.wt. proved to be hepatotoxic and nephrotoxic in camels. The study showed an excellent potential for future investigations to determine the effect of chronic administration of smaller doses of gliotoxin to camels which may occur under natural exposure.
The author is thankful to the deanship of Scientific Research for financial support.